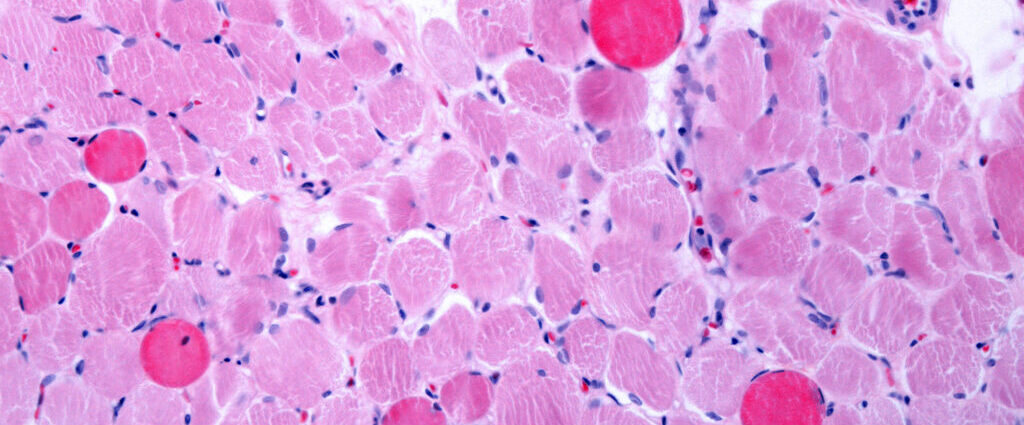
Entrada Therapeutics’ next-generation drug for Duchenne muscular dystrophy disappointed in an early trial, raising questions about the company’s competitiveness in an increasingly crowded field.
Entrada is one of a group of companies developing new exon-skipping drugs. These medicines are designed to help patients with certain mutations produce shortened but still functional forms of dystrophin, the protein missing in Duchenne.
The first such drug, from Sarepta Therapeutics, had only marginal effects on protein production but was approved in 2016 under immense pressure from patient advocates. Since then, scientists have devised ways of redesigning these molecules to better infiltrate muscle cells, leading to vastly higher dystrophin levels.