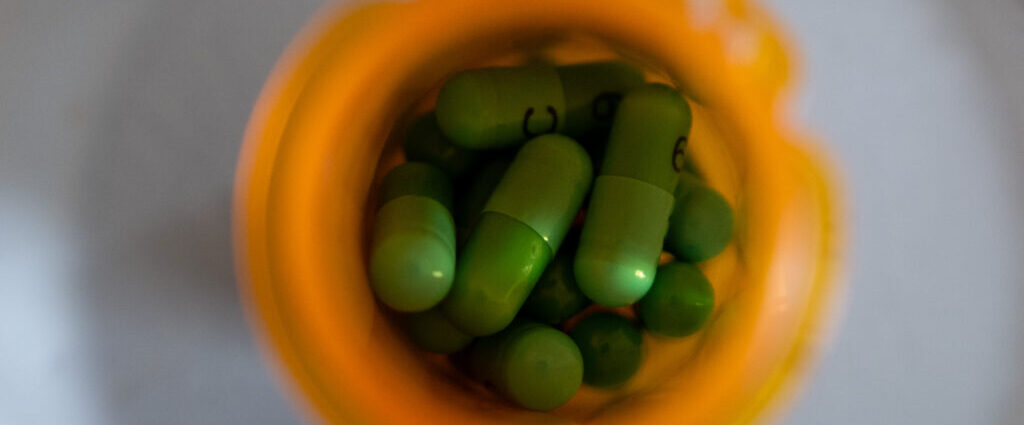
PFAS is still in some infant formula
Get your daily dose of health and medicine every weekday with STAT’s free newsletter Morning Rounds. Sign up here.
Too much news yesterday, so here’s a link to a late-breaking story that the FDA has chosen Vinay Prasad’s interim successor.
STAT+: Pharmalittle: We’re reading about obesity drugs and a compounding list, an AstraZeneca setback, and more
And so, another working week will soon draw to a close. Not a moment too soon, yes? This is, you may recall, our treasured signal to daydream about weekend plans. Our agenda: promenading with the official mascots, manicuring the Pharmalot grounds, and escorting Mrs. Pharmalot to a supper club. We also hope to find time for yet another listening party, where the rotation will likely include this, this, this, this, this. And what about you? As we like to remind folks, this is a fine time to enjoy the great outdoors. Hiking, strolling, dipping your toe in the water — take your pick. Of course, if the weather fails to cooperate, you could catch up on your reading, binge-watch on the telly, or tidy up around the castle. The possibilities are endless, yes? Well, whatever you do, have a grand time. But be safe. Enjoy, and see you soon….
The U.S. Food and Drug Administration proposed excluding the active ingredients in wildly popular obesity and diabetes drugs from a list of substances that can be used for making certain compounded versions, STAT writes. The agency determined there is no “clinical need” to allow large compounding facilities to make large quantities of medicines from semaglutide, the ingredient in Novo Nordisk’s Wegovy weight loss and Ozempic diabetes treatments, as well as tirzepatide, the ingredient in Eli Lilly’s Mounjaro diabetes drug and Zepbound obesity medicine. The decision singled out these compounders after growing controversy over their role in making weight loss treatments available over the past few years. In explaining its decision — which is a victory for Novo and Lilly and will rework choices for consumers — the FDA explained these compounders no longer meet the legal requirements to market their products.
The FDA named Katherine Szarama as the acting director of the Center for Biologics Evaluation and Research, which regulates vaccines, gene therapies, and the blood supply, STAT notes. She is replacing Vinay Prasad, who left the agency on Thursday after a tumultuous tenure during which he issued a series of controversial decisions on rare disease drugs and vaccines. FDA Commissioner Marty Makary said in March that Prasad would return to the University of California, San Francisco. Szarama joined the FDA at the end of last year to serve as Prasad’s deputy. It is not clear whether Szarama will serve permanently in the role. Government and industry sources previously indicated that Houman Hemmati, an ophthalmologist, biopharma executive, and frequent Fox News contributor, was a top candidate for the role.
STAT+: Axsome wins FDA nod for Alzheimer’s agitation
Want to stay on top of the science and politics driving biotech today? Sign up to get our biotech newsletter in your inbox.
The FDA named Katherine Szarama acting head of CBER as it continues to search for a permanent leader. Julia Vitarello is launching a new effort to scale bespoke medicines after her first startup faltered.
And Matt Herper reflects on the legacy of genomics pioneer J. Craig Venter, whose vision helped shape modern biotech.
Opinion: The psychedelic revolution is leaving behind people of color
Flanked by one of psychedelics’ biggest celebrity cheerleaders, Joe Rogan, and a troupe of MAHA loyalists, President Trump recently signed an executive order aimed at accelerating psychedelic access for clinical research and treatment.
Use of naturally occurring and synthetic hallucinogens traces back to the Neanderthals. Yet these substances have long been a pariah in mainstream medicine, written off as “club drugs” with little to no clinical value (or worse, downright negative effects).
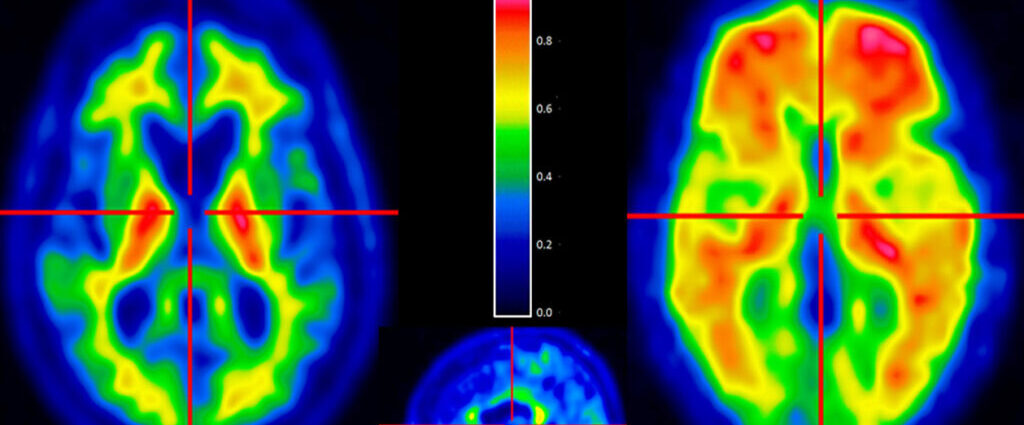
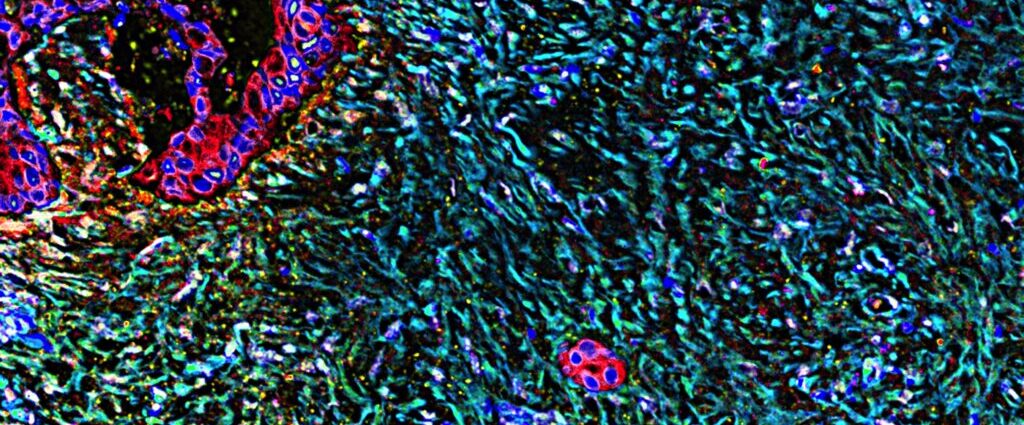
‘Forever chemicals’ in baby formula? Scientists unpack FDA results
The big takeaway from a new government survey of infant formula is that the U.S. supply is largely safe. But experts and health officials say there are still steps that can be taken to make a product consumed by two-thirds of infants in the U.S. even safer.
One noteworthy finding from the Food and Drug Administration’s testing of 312 formula samples concerned per- and polyfluoroalkyl substances, or PFAS, also known as “forever chemicals.” The FDA detected five PFAS in the samples it tested, with the most common one — PFOS — found in half of all samples. Of those samples, the vast majority (95%) contained less than 2.9 parts per trillion (ppt) of PFOS.
What, exactly, does that mean? The FDA analysis doesn’t explain the PFAS results in much detail. But parents are bound to wonder, given that higher levels of exposure to PFAS, man-made chemicals used in products like nonstick cookware and stain-resistant clothing and rugs, have been linked to conditions including higher cholesterol, kidney and testicular cancer, and reduced vaccine efficacy.
STAT+: Her daughter Mila got a bespoke medicine. Now she’s starting a new biotech to make more
ROME — Julia Vitarello, whose daughter Mila eight years ago received a bespoke medicine designed for her particular disease-causing mutation, said this week that she is in the process of starting a new company to try to create these individualized therapies at scale.
Vitarello’s previous effort, called EveryONE Medicines, recently folded in part because new Food and Drug Administration guidance encouraging the development of customized therapies did not go far enough in creating a pathway to satisfy EveryONE’s investors, Vitarello said.
Now Vitarello and collaborators are looking for new funders.
Opinion: America is worrying about fertility again. But it’s not really about families
America is worried about fertility rates — again.
Coverage of the country’s declining birth rate reflects widespread unease: Families are struggling, young adults are delaying or forgoing parenthood, and the future labor force feels uncertain. These concerns are rooted in real social and economic challenges. Childcare is unaffordable, housing is out of reach for many, health care access is precarious, and paid parental leave is still not guaranteed. Only now, as these pressures have produced sustained fertility declines, have calls for expanded material supports gained real political traction.
STAT+: OxyContin maker Purdue Pharma set to dissolve after judge approves its criminal sentence
NEWARK, N.J. — OxyContin maker Purdue Pharma is set to be dissolved and replaced by a company focused on the public good by the week’s end, as a massive legal settlement resolving thousands of lawsuits takes effect.
A federal judge on Tuesday delivered a criminal sentence to the company to resolve a Department of Justice probe — a last necessary step to clear the way for the settlement.
U.S. District Judge Madeline Cox Arleo made her decision after listening to hours of impact statements from people who lost loved ones or struggled with addiction themselves and requested she reject the negotiated sentence. While she didn’t go that far, she said she sympathized with people who bore the brunt of an epidemic linked to more than 900,000 deaths in the U.S. since 1999.
Remembering J. Craig Venter: a relentless scientist who changed biotech — and was all too easily misunderstood
J. Craig Venter, a scientist whose relentless ambition helped turn genetics from an artisanal trade into an industrialized information machine, died Wednesday at 79. The cause was side effects of a cancer treatment.
Along the way, he did things that can only be described as really cool. He raced against a government-funded project to sequence the first human genome, grabbing headlines around the world; traveled the ocean in his sailboat collecting genetic information about sea life; and removed a bacterium’s genome and rebooted the organism with an identical set of genes he and his team had synthesized. He drove fast cars, drank red wine, and pissed people off.
OSHA Issues New Guidance on Heat Stress Protection for Outdoor and Warehouse Workers as Summer Approaches
By JBizNews Desk — April 30, 2026
The Occupational Safety and Health Administration released updated guidance late Wednesday urging small businesses with outdoor or warehouse operations to implement mandatory heat-stress prevention plans ahead of rising summer temperatures, citing increased claims linked to extreme heat events. The after-close advisory, sent to trade associations and small-employer networks, emphasizes paid rest breaks, hydration stations, and training — measures many smaller operators say will add to already elevated labor and insurance costs tracked throughout the day.
For small construction firms, landscapers, delivery services, and warehouse operators, the new expectations could require schedule adjustments or equipment investments at a time when hiring remains cautious and consumer spending is restrained.
Key Requirements in the New Guidance
• Mandatory 15-minute paid rest breaks every two hours when heat index exceeds 90°F.
• Free provision of water, shade structures, and training for supervisors and workers.
• Recommended written heat-illness prevention plans for businesses with 10 or more outdoor employees.
Economists described the guidance as a necessary but costly step for small employers already navigating multiple pressures, with Diane Swonk, chief economist at KPMG, noting that as diesel’s cost advantage erodes amid volatile fuel prices, fleets and small operators are increasingly open to electric alternatives but now face added compliance costs; Heather Long, chief economist at Navy Federal Credit Union, pointed out the ripple effects for everyday workers and businesses as cautious consumer spending weighs on growth; Oliver Allen, senior U.S. economist at Pantheon Macroeconomics, emphasized that this reflects broader trends of regulatory support for worker safety in a high-cost environment; Nicole Bachaud, economist at ZipRecruiter, added that the measures could encourage more selective hiring and training investments; and Gina Bolvin, president of Bolvin Wealth Management Group, advised small-business clients to implement low-cost compliance steps early to avoid larger insurance claims or fines.
Outlook
The OSHA heat-stress guidance arrives as small businesses prepare for another summer of elevated operational demands. For Main Street operators and their workers, the advisory underscores the growing intersection of safety, labor, and cost management. Tomorrow’s small-business labor updates will reveal how quickly employers adapt these recommendations.
JBizNews Desk
© JBizNews.com. All rights reserved. This article is original reporting by JBizNews Desk. Unauthorized reproduction or redistribution is strictly prohibited.
STAT+: Katherine Szarama named acting director of FDA’s vaccines and biologics center
WASHINGTON — The Food and Drug Administration has named Katherine Szarama as the acting director of the Center for Biologics Evaluation and Research, which regulates vaccines, gene therapies, and the blood supply.
A Health and Human Services official confirmed the move, which was first reported by Politico, to STAT.
She is replacing Vinay Prasad, who left the agency on Thursday after a tumultuous tenure during which he issued a series of controversial decisions on rare disease drugs and vaccines. FDA Commissioner Marty Makary said in March that Prasad would return to the University of California San Francisco.
STAT+: In her own words: Surgeon general nominee Nicole Saphier expresses enthusiasm and caution for MAHA
Now that Casey Means is no longer the Trump administration’s choice for Surgeon General, attention is turning to the third nominee for the position.
Nicole Saphier, whose candidacy was announced Thursday, is a licensed physician — unlike Means, whose license lapsed. A radiologist at the Memorial Sloan Kettering Cancer Center, Saphier (pronounced SAA-fire) is director of breast imaging at MSK Monmouth in New Jersey. She may be more widely known as a regular contributor to Fox Business, where she has said that the overwhelming majority of “good research” disputes the notion that vaccines are linked to autism, but has expressed an openness to alternative childhood vaccine schedules.
Saphier has weighed in on many other concerns shared by the Make America Healthy Again movement promoted by health secretary Robert F. Kennedy Jr., agreeing with Kennedy on some but also clearly questioning on others. In her own words, here are her views on vaccines, peptides, Tylenol in pregnancy, dietary guidelines, breast cancer, and also, Casey Means.
STAT+: Hair-raising trial results, and Servier’s M&A wishlist
Why are investors excited about hair loss drugs? Will artificial intelligence make clinical trials run more smoothly? And how does a nonprofit pharma company compete in the M&A arena?
We get into all that and more on this week’s episode of “The Readout LOUD,” STAT’s weekly biotech podcast.
Veradermics CEO Reid Waldman joined us to discuss his company’s data, and why hair loss is such a trendy topic in biotech. Then, Servier Pharmaceuticals CEO David Lee joined us to discuss the company’s acquisition of Day One Biopharmaceuticals. The hosts also discussed the latest news in biotech.
STAT+: FDA wants to exclude weight loss drugs from a compounding list
The Food and Drug Administration has proposed excluding the active ingredients in wildly popular obesity and diabetes drugs from a list of substances that can be used for making certain compounded versions.
The agency determined there is no “clinical need” to allow large compounding facilities to make large quantities of medicines from semaglutide, the ingredient in Novo Nordisk’s Wegovy weight loss and Ozempic diabetes treatments, as well as tirzepatide, the ingredient in Eli Lilly’s Mounjaro diabetes drug and Zepbound obesity medicine.
The decision singled out these compounders, which are known as 503B facilities in regulatory parlance, after growing controversy over their role in making weight loss treatments available over the past few years. In explaining its decision — which is a victory for Novo and Lilly and will rework choices for consumers — the FDA explained these compounders no longer meet the legal requirements to market their products.
Trump drops Casey Means as surgeon general nominee, opts for radiologist Nicole Saphier
WASHINGTON — The Trump administration has dropped its nomination of Casey Means to be the next surgeon general, opting instead for Nicole Saphier, a radiologist and Fox News contributor.
The move represents a blow to Make America Healthy Again movement’s months-long push to get Means confirmed. But Saphier shares some commonalities with the MAHA movement, including an interest in personal wellness and a skepticism of vaccine mandates.
STAT+: As artificial intelligence shows off diagnostic chops, scientists reckon with the way forward
Getting a paper published in Science is a highlight of many researchers’ careers. But for internist and clinical artificial intelligence researcher Adam Rodman, it’s also been a source of some agita.
On Thursday, Rodman and his colleagues published a compilation of experiments, including one using real-world data from a Boston emergency department, that show a large language model from OpenAI can outperform physicians in case-based diagnostic and clinical reasoning evaluations. To Rodman, the paper’s co-senior author, it’s a response to a gauntlet thrown down in Science in 1959. That paper “described how you would know that a clinical decision support system was capable of doing diagnosis better than humans,” he said. “And they can do it.”
But as generative AI tools like chatbots are heavily marketed — both to patients and clinicians — it makes him worried that the science experiments, all based on simulated and historical cases, will be misconstrued as proof of AI’s safety and efficacy when used to treat real patients.
STAT+: DOJ, Elevance spar over access to top exec in Medicare Advantage fraud case
One of the biggest Medicare Advantage fraud cases has hit a snag, with government officials saying they don’t have access to a key witness.
Elevance Health is blocking the deposition of Peter Haytaian, an outgoing executive who previously oversaw the health insurer’s Medicare Advantage plans, Department of Justice attorneys said this week in a letter to a judge. The letter also revealed how Elevance announced Haytaian’s departure right after DOJ first asked to depose him.
The dispute underscores the high stakes of the lawsuit, which the DOJ initiated in 2020. It also adds to the wide-ranging scrutiny facing Elevance’s private Medicare plans for seniors, which generated more than $40 billion of revenue last year.
STAT+: Five-way obesity drug is super effective — in mice
Want to stay on top of the science and politics driving biotech today? Sign up to get our biotech newsletter in your inbox.
Move over, GLP-1s. A preclinical next-gen obesity drug is stacking five mechanisms (in mice). Also, the NIH funding climate is rougher than ever, Novartis is reshoring more of its manufacturing, and another Alzheimer’s program is scuttled.
The need-to-know this morning
- More pharma earnings: Merck, Bristol Myers Squibb and Eli Lilly
- Notable biotech earnings: Alnylam.
- UniQure said it will submit a marketing application for its Huntington’s disease gene therapy to U.K. regulators in the third quarter.
- Craig Venter, scientist, genomic pioneer and entrepreneur, died.
It’s far harder to get an NIH grant these days
The odds of landing an NIH grant have cratered to near-unsustainable levels. Only 13% of applications were funded last year, STAT’s Anil Oza writes, with even top-scoring proposals no longer having good odds. Now, researchers are scrambling to survive in a system that’s become both hypercompetitive and opaque.
STAT+: Democrats go hard on health care affordability
You’re reading the web edition of D.C. Diagnosis, STAT’s twice-weekly newsletter about the politics and policy of health and medicine. Sign up here to receive it in your inbox on Tuesdays and Thursdays.
In the health secretary’s latest podcast, Mike Tyson and RFK Jr. compared their diets in the clink, and Iron Mike blamed prison violence on ultra-processed food. Share news tips and prison experiences at John.Wilkerson@statnews.com or John_Wilkerson.07 on Signal.
Lower health care costs for everything, everywhere
Senate Minority Leader Chuck Schumer (D-N.Y.) outlined health care policy principles that call for lowering costs for everything, everywhere.
STAT+: Cigna will exit ACA individual markets next year, adding to tumult for patients
Cigna is pulling out of the Affordable Care Act’s individual marketplaces in 2027 to focus on more promising parts of its business, another exit from what’s become a tumultuous piece of the insurance industry.
Executives announced the decision on Cigna’s first quarter earnings call on Thursday, in which the company unveiled better-than-expected financial results, including $1.7 billion in profit, and a bumped-up earnings forecast for the year. Cigna currently has 369,000 ACA members across 11 states, a small portion of its 18.3 million total members.
Brian Evanko, Cigna’s chief operating officer, said on the call that Cigna did not take the decision lightly. He said it was driven by two main factors: Cigna did not see a way to meaningfully grow that business and cutting it would free Cigna to focus on priorities like its Evernorth specialty and care services division, its pharmacy benefits division, and its flagship employer plan business.
STAT+: Hinge has a new FDA-cleared migraine device
You’re reading the web edition of STAT’s Health Tech newsletter, our guide to how technology is transforming the life sciences. Sign up to get it delivered in your inbox every Tuesday and Thursday.
Good morning health tech readers!
Today, updates from large virtual care companies. Plus: A low-tech way to boost cancer screenings.
STAT+: Pharmalittle: We’re reading about a Supreme Court hearing on ‘skinny labels,’ AstraZeneca U.K. expansion, and more
Rise and shine, everyone, another busy day is on the way. Sadly, gray skies are hovering over the Pharmalot campus, but our spirits remain sunny, nonetheless. And why not? As the Morning Mayor suggested to us long ago, “Every day should be unwrapped like a precious gift.” While you tug on the ribbon, we will fire up the trusty kettle for a cuppa elderberry hibiscus. Of course, you are invited to join us. Remember, no prescription is required. This is strictly cash money. Meanwhile, here are a few items of interest. Hope you have a meaningful and productive day and, of course, do stay in touch. …
AstraZeneca will resume expansion of its research and development operations in a Cambridge, U.K. facility, the first such project to be revived following the trade deal with the U.S. that included measures to lift U.K. spending on medicines, The Financial Times writes. Pascal Soriot, AstraZeneca’s chief executive, said on Wednesday that the drugmaker would invest $400 million to complete the Rosalind Franklin building that it put on hold last year, and build a new laboratory in the town of Macclesfield that would use “digital and data tools to advance drug development.” The company was one of several drugmakers that postponed or cancelled U.K. investments over disagreements with the government about increased medicines spending. As part of a trade deal agreed with the Trump administration, the U.K. agreed to lift the thresholds it uses to determine whether medicines are cost-effective for use on the National Health Service.
After months of anticipation, U.S. Supreme Court justices heard arguments about a long-standing tactic used by generic companies to carve out a distinct market for a medicine, and did not appear inclined to alter legal standards for the maneuver, STAT explains. At issue is skinny labeling, which refers to moves by generic companies that seek regulatory approval to market a drug for a specific use, but not other patented uses for which a brand-name medicine is prescribed. For instance, a generic drug could be marketed to treat one type of heart problem but not another. In doing so, the generic company seeks to avoid lawsuits claiming patent infringement. The court heard arguments concerning a case involving Amarin, which markets a drug called Vascepa for treating different heart problems, and Hikma Pharmaceuticals, one of the largest generic manufacturers.
STAT+: What does Akeso’s primo plenary spot say about its survival data?
This is the online version of Adam’s Biotech Scorecard, a subscriber-only newsletter. STAT+ subscribers can sign up here to get it delivered to their inbox.
I’m suffering from spring allergies + a nasty head cold this week. Not fun.
Mystery cloaks an ASCO plenary lung cancer survival result
The runup to next month’s annual meeting of the American Society of Clinical Oncology has started, and a good bit of the Wall Street chatter and speculation is centered on the Akeso-Summit Therapeutics drug ivonescimab.
Smog from wildfires increasingly harmful, data show
Get your daily dose of health and medicine every weekday with STAT’s free newsletter Morning Rounds. Sign up here.
Good morning. If you feel strongly about health care v. healthcare, now’s your chance to weigh in. (We’ve already heard from nearly 100 readers!)
STAT+: New obesity tool aims to predict risk of 18 serious complications
Body mass index has its limitations, but for now it’s the metric medicine often defaults to when predicting weight-related health problems. A new tool promises to better define who’s at risk for obesity complications, based on measures that include BMI but also family history, diet, current illness, and socioeconomic factors culled from medical records.
One aim of the research is to better understand who’s a candidate for an obesity drug, often prescribed based on BMI alone or BMI in combination with another disease. Over time, GLP-1 medications, whose initial target was type 2 diabetes, have revealed their power to ease cardiovascular disease, kidney disease, liver disease, sleep apnea, and osteoarthritis, in addition to promoting significant weight loss. But discerning who’s the best fit for the costly, lifelong treatment has been uncertain.
“We really wanted to have an integrated model that enables us to look at not one, but 18 different obesity-relevant complications,” Claudia Langenberg, co-author of a study about the new model published Thursday in Nature Medicine, said in a media briefing Tuesday. She is director and professor of medicine and population health at Precision Healthcare University Research Institute of Queen Mary University of London.
Opinion: Congress must hold RFK Jr. accountable after hearings
Our former colleagues in Congress recently heard from Health and Human Services Secretary Robert F. Kennedy Jr. for the first time in more than half a year.
Several congressional committees held hearings with Kennedy as they examined the Trump administration’s budget priorities. It was a key opportunity for lawmakers to build on the administration’s significant health policy achievements — and for the most part, they seized it. While Democrats were predictably critical of the secretary’s every move, Republicans wisely pushed back on the administration’s proposal to reduce funding for the National Institutes of Health. And with good reason. Sustained NIH funding underpins the research and development pipeline that makes vaccines possible at a time when voter polling shows Americans want leaders that support vaccine access.
Securing NIH awards is getting more competitive — and confusing
The likelihood of snagging National Institutes of Health grants has plunged to historic lows, forcing frustrated academic researchers to resort to a variety of tactics to try to obtain funding and, in some cases, keep their jobs, according to a nationwide STAT survey and follow-up interviews with respondents.
NIH data show that securing research awards has become more competitive under the second Trump administration than ever before, and more unpredictable. Just 13% of applications were funded in the past fiscal year, and even top-rated proposals aren’t a sure thing.
Opinion: Are international medical graduates taking residency spots from Americans? We did the math
The 2026 Main Residency Match was the largest in the program’s 74-year history. With 48,050 active applicants, 41,482 residency training positions were filled.
Among the celebrations, however, Match Day announcements triggered a surge of posts on X and other social media platforms calling for new restrictions on international medical graduates (IMGs) in the match, claiming that U.S. medical graduates (USMGs) had been displaced. These calls came from a range of voices: anti-immigration commentators and political figures with no medical background as well as physicians channeling workforce frustration into criticism of non-U.S. IMGs. Critics found headshots of incoming residents, posted by programs to celebrate their matches, and recirculated without consent to fuel this narrative. Certain policymakers and social media figures with large followings have amplified these claims and are actively pursuing legislation to restrict visa sponsorship for internationally trained physicians.
HHS appeals ruling that stalled many of Kennedy’s vaccine policy changes
The Department of Justice is appealing a ruling by a federal judge which stalled many of health secretary Robert F. Kennedy Jr.’s changes to federal vaccine policy.
The appeal, filed Wednesday, is the latest twist in the closely watched lawsuit filed by the American Association of Pediatrics along with several other professional organizations and several anonymous women. The plaintiffs argued that in reconstituting a federal vaccine advisory committee and changing the childhood vaccine schedule, Kennedy’s health department violated the Administrative Procedures Act, which governs how federal agencies consider and implement policy changes.
STAT+: Supreme Court justices appear to signal ‘skinny labeling’ can survive
After months of anticipation, Supreme Court justices heard arguments about a long-standing tactic used by generic companies to carve out a distinct market for a medicine, and did not appear inclined to alter legal standards for the maneuver.
At issue is skinny labeling, which refers to moves by generic companies that seek regulatory approval to market a drug for a specific use, but not other patented uses for which a brand-name medicine is prescribed. For instance, a generic drug could be marketed to treat one type of heart problem but not another. In doing so, the generic company seeks to avoid lawsuits claiming patent infringement.
This tactic has been a key tool for generic companies ever since the Hatch-Waxman Act was signed into law more than four decades ago. The law established the mechanisms by which generic drugs can more readily enter the marketplace. And skinny labeling is one way that Congress attempted to foster more competition and benefit consumers.
STAT+: Why conversations around health AI may be evolving beyond hype
You’re reading the web edition of STAT’s AI Prognosis newsletter, our subscriber-exclusive guide to artificial intelligence in health care and medicine. Sign up to get it delivered in your inbox every Wednesday.
My phone blew up while I was on vacation last week: The Associated Press Stylebook announced “health care” should actually be one word, “healthcare.” STAT is still deciding which we should use. Which do you prefer? You can weigh in here.
(Will my newsletters technically be shorter if we switch to “healthcare”? Food for thought.)
Maybe now we can have a real conversation about AI in health care
In his recent video “The People Do Not Yearn for Automation,” The Verge editor-in-chief Nilay Patel explains “software brain” — thinking about the world as a series of databases that are easily manipulated to solve problems — and why that is creating a disconnect between the AI world and everyone else.
The AI world thinks that AI really can solve the world’s ills and thus anti-AI sentiment is just a marketing problem. But people who are trying to slow AI adoption have legitimate concerns about the tradeoffs and performance of the technology that software-brained people are dismissing, he says.
Infant formula largely safe from heavy metals, FDA finds
New contamination testing results from the Food and Drug Administration confirm the safety of infant formula in the U.S., the agency said Wednesday.
The FDA tested 312 samples from 16 infant formula brands for contaminants like heavy metals, pesticides, and the “forever chemicals” known as per- and polyfluoroalkyl substances, or PFAS. The vast majority had undetectable or very low levels of contaminants, the agency said, with levels of lead, mercury, cadmium, and arsenic coming in below federal requirements for drinking water across all samples.
STAT+: Pharmalittle: We’re reading about the FDA speeding up trials, a Supreme Court hearing on ‘skinny labels,’ and more
Top of the morning to you. The middle of the week is upon us and, since you made it this far, why not forge ahead? After all, there is always light at the end of the proverbial tunnel. You never know what you may accomplish. So please join us as we celebrate this notion with a cup or three of delicious stimulation. Our choice today is chocolate raspberry. Meanwhile, we have assembled the latest menu of tidbits to help you along. So please dig in. Have a smashing day, and please feel free to forward any secrets you come across. Our “in basket” is always open. …
The U.S. Food and Drug Administration announced efforts to make clinical trials more efficient, starting by reviewing data in real time from trials conducted by AstraZeneca and Amgen, STAT writes. The agency also asked the public to weigh in on a potential pilot program to work with companies that use AI to enhance safety monitoring and medication dose selections, identify safety signals, and improve patient recruitment in clinical trials. The trials will rely on a real-time data platform built by Paradigm Health, and the goal is to cut down on the time regulators and companies spend sending data back and forth. FDA Commissioner Marty Makary said at a press conference that agency reviewers will be able to view safety signals and clinical endpoints via Paradigm’s platform.
Pfizer settled patent disputes with three generic drugmakers over its blockbuster heart drug Vyndamax, effectively extending its patent protection until 2031 and delaying cheaper copies from entering the market, Reuters says. The deals resolve patent infringement lawsuits against Dexcel Pharma, Hikma Pharmaceuticals, and Cipla in Delaware federal court over Pfizer’s oral drug Vyndamax. A trial over the patent had started this week. Pfizer sold nearly $6.4 billion of Vyndamax and related drugs, which treat a serious heart condition called transthyretin amyloid cardiomyopathy, in 2025. The settlements extend U.S. patent protection for Vyndamax until June 1, 2031, subject to other pending litigation. The company had previously expected a sharp drop in U.S. revenue for the drug in 2029 but now expects sales to hold relatively steady from 2028 through mid-2031.
STAT+: A patent win for Pfizer and BridgeBio
Want to stay on top of the science and politics driving biotech today? Sign up to get our biotech newsletter in your inbox.
Good morning. Several biotech leaders were named in this list of the most influential Bostonians. Is there anyone you think should have made the list who didn’t? Let me know your thoughts.
The need-to-know this morning
- The Italian pharma company Chiesi is buying KalVista Therapeutics for $1.9 billion, adding an approved treatment for a genetic swelling disorder to its rare-disease portfolio. The deal values KalVista at $27 per share, or a 40% premium to its Tuesday closing stock price. The KalVista medicine, called Ekterly, is a pill used to treat acute swelling attacks in people with hereditary angioedema.
- Biogen, AstraZeneca, Regeneron Pharma, Abbvie and GSK reported first-quarter earnings.
FDA plans to review AZ, Amgen trials in real time
In an effort to get drugs on the market faster, the FDA announced yesterday it will start reviewing trial data in real time, starting with oncology studies conducted by AstraZeneca and Amgen.
UN Report Finds Workplace Stress Linked to 840,000 Deaths Annually
A sweeping new report from the International Labour Organization (ILO) has quantified the global toll of toxic workplace conditions, finding that more than 840,000 people die each year from health issues directly linked to stress, burnout, harassment, and excessive working hours.
The report marks one of the most comprehensive global assessments of workplace-related psychosocial risks, identifying cardiovascular disease and mental health disorders — including suicide — as the primary causes of these deaths.
“These are not isolated issues — they are systemic,” said Manal Azzi, team lead on occupational safety and health policy at the ILO. “Psychosocial risks are now one of the defining challenges of modern work.”
The data paints a stark picture. The report estimates nearly 45 million disability-adjusted life years lost annually, capturing not just mortality but years of productivity lost to illness and disability. The economic cost is equally significant, amounting to roughly 1.37% of global GDP, or about $1.6 trillion annually.
In Europe alone, workplace stress-related conditions account for more than 112,000 deaths each year, with a measurable drag on economic output. The findings underscore how workplace environments have evolved — and in many cases deteriorated — amid increasing demands, job insecurity, and blurred boundaries between work and personal life.
The most common health outcomes identified include depression, anxiety, chronic fatigue, and sleep disorders. These conditions often trigger secondary health risks, including substance use, obesity, and hypertension.
For employers, the implications are increasingly financial. “Companies that ignore workplace wellbeing are effectively taking on hidden liabilities,” said one labor economist. Rising absenteeism, turnover, and healthcare costs are directly tied to workplace conditions.
The ILO is calling for systemic reforms, including improved job design, stronger employee protections, and proactive mental health support systems. The report also urges companies to treat workplace wellbeing as a core operational priority rather than a secondary human resources issue.
As businesses navigate a shifting labor market, the findings reinforce a growing reality: workforce health is not just a social issue — it is a central pillar of long-term economic performance.
JBizNews Desk
State Farm Faces Wave of Lawsuits Alleging Systematic Denial of Hail Damage Claims
State Farm, the largest home insurer in the United States, is facing an expanding legal battle spanning multiple states, with hundreds of homeowners alleging the company engaged in a coordinated effort to deny or underpay legitimate claims tied to hail and wind damage — even when those claims fell within the terms of their policies.
The litigation, which includes more than 600 active cases in Oklahoma alone, centers on accusations that the insurer implemented internal practices designed to limit costly roof replacement payouts. Plaintiffs argue that beginning around 2020, State Farm quietly introduced new internal standards — particularly in Texas before expanding elsewhere — that effectively narrowed the definition of what constitutes covered damage.
According to court filings and attorneys involved in the cases, one of the primary tactics involved requiring proof of “functional damage,” a threshold that critics say is not outlined in standard policy language. By applying these criteria, plaintiffs allege, the company was able to deny claims outright or significantly reduce payouts, leaving homeowners responsible for repairs they believed were covered.
The dispute has escalated to the level of state government. Oklahoma Attorney General Gentner Drummond has joined one of the lawsuits, alleging what he described as a “systematic scheme” to deny valid claims. His involvement has amplified national attention on the case, particularly as homeowners across the country grapple with rising insurance costs and growing uncertainty about coverage reliability.
State Farm has pushed back strongly against the allegations. In public statements, the company has maintained that it evaluates claims based strictly on policy terms and individual circumstances. “We pay what we owe under the policy,” the insurer said, rejecting claims of any coordinated misconduct and warning that inflated claims from contractors and legal firms can distort the system.
Still, critics point to a pattern of multimillion-dollar confidential settlements in some cases as evidence of potential exposure. Legal analysts say the volume and consistency of the lawsuits suggest deeper systemic questions about claims practices across the industry.
The timing is particularly sensitive. The Insurance Information Institute estimates that severe storms caused $51 billion in insured losses last year, driven in part by increasing frequency and intensity of extreme weather events. At the same time, insurers have raised premiums sharply or withdrawn from high-risk markets altogether.
“This is happening at a moment when homeowners are already under enormous pressure,” said one industry analyst. “If trust in claims payouts erodes, that undermines the entire insurance model.”
The outcome of the litigation could have far-reaching implications — not only for State Farm, but for how insurers nationwide define, evaluate, and pay storm-related claims in an era of rising climate risk.
JBizNews Desk
Food safety experts warn of USDA brain drain
Get your daily dose of health and medicine every weekday with STAT’s free newsletter Morning Rounds. Sign up here.
Morning. In the same way that Athena orchestrated the logistics around Telemachus’s journey in the first three books of “The Odyssey,” I hope some gods out there are coordinating a “Survivor” season 50 win for Cirie Fields. She’s earning it, but I wouldn’t mind some divine intervention to make sure.
STAT+: Google clinical director says AI can be a ‘bridge’ for people having a mental health crisis
As Google faces pressure to take greater accountability for the mental health impacts of its artificial intelligence products, the company’s clinical director Megan Jones Bell welcomed the challenge of making artificial intelligence helpful to people who come to its Gemini chatbot with a mental health crisis.
“It can seem sometimes like shutting something down is a way of preventing harm,” Jones Bell told STAT. “We believe that making our product experience safer and more helpful and strengthening that bridge to support it is the more effective path to support mental health for the most people.”
Google recently made updates to its Gemini app so that it more prominently features connections to crisis hotlines when it detects a person may be at risk of self harm. In conversations about mental health, the AI will frequently point people to outside resources — but the bot doesn’t disengage, reminding a user, for example, that “I’m here to listen.”
STAT+: A giant question looms over GOP’s new Medicaid work requirements
WASHINGTON — Democrats and Republicans are at odds over a consequential health care policy embedded in last year’s tax cut law.
Democrats say the law’s Medicaid work requirements will create red tape that leads to people losing their coverage. Republicans say the measure encourages work.
But because the law doesn’t require states to report on the implementation of this massive change to Medicaid, we may never get a detailed account of how the policy is working.
Opinion: Why do discussions about ‘brain health’ ignore mental illness?
Governments, industry, and philanthropies are investing in neuroscience at an unprecedented scale, and the ambition behind this impetus is a noble one: to reduce the growing burden of brain diseases and extend healthy cognitive life. We fully support this movement’s push for “brain health” to mirror successful frameworks established for cancer and heart health that prioritize early screening and aggressive preventive treatments, making it possible to act before irreversible damage sets in.
Even as this agenda gains momentum, however, a critical blind spot is emerging. As governments refocus their policies to tackle conditions like Alzheimer’s disease and other neurodegenerative disorders, mental illness is often being sidelined as a secondary concern rather than as a primary component of brain health. This artificial divide is a scientific and a strategic error.
STAT+: Eli Lilly enlists AI startup for next-generation gene editors
Eli Lilly struck a deal Tuesday to develop new forms of gene editors potentially capable of inserting entire genes into patients.
The collaboration, with artificial intelligence-focused biotech Profluent, is sparse on details, including the number of programs the two companies would work on, the types of diseases they’ll pursue, or how much Lilly was paying upfront. But if every one of its efforts works out, Lilly would pay Profluent $2.25 billion in milestones payments.
The deal is part of a larger push by Lilly into gene editing. The big pharma, flush with record revenues from its obesity and diabetes drugs, has opened a new genetic medicine center in Boston and bought up a series of gene editing or gene therapy companies over the last few years.
Predicting consequences of new hepatitis B vaccine recs
Get your daily dose of health and medicine every weekday with STAT’s free newsletter Morning Rounds. Sign up here.
Good morning. The other night I watched a shocking episode of “The Vampire Diaries.” A series of cursed, ghost-like hallucinations attempt to convince a teen vampire to end her own life using some disturbingly coercive, cogent arguments. Ultimately, the character is saved. And while this episode aired more than a decade ago, I was surprised by how many parallels there were to current debates about the risks of AI chatbots and people in mental health crises.
STAT+: Pharmalittle: We’re reading about warnings over Trump’s MFN plan, Purdue Pharma sentencing, and more
Rise and shine, everyone, another busy day is on the way. And it is getting off to a good start here on the Pharmalot campus, where clear blue skies and pleasant breezes are greeting us. Who could ask for anything more? Actually, we could — it is time to reheat the kettle for another cuppa stimulation. Our choice today is apple hibiscus. And here is a helpful tip — a teaspoon of honey enhances the flavors splendidly. Of course, you are invited to join us. For the full experience, we are now hawking replicas — take a look. Meanwhile, here are a few tidbits to help you along. As always, do keep in touch. We appreciate feedback, criticism, and tips. …
Novartis chief executive officer Vas Narasimhan warned that the U.S. drug pricing policy under President Trump poses a “very difficult situation” and the reality will soon catch up with both drugmakers and patients, CNBC tells us. He maintained that Novartis is focused on getting European and Japanese governments to quickly change how they reward innovation, and added that if this does not happen, then novel medicines might see delayed entry into these markets and patients won’t have access to the drugs. The most-favored-nation drug pricing policy, or MFN, implemented by Trump last year means that prices in the big and lucrative U.S. market are tied to prices in comparably wealthy countries. Trump has made lower drug prices for Americans a priority and has long criticized what he calls “foreign nations freeloading on American-financed innovation.”
Japan risks missing out on new blockbuster treatments due to a drug pricing scheme being adopted in the U.S., Nikkei Asia writes. The Trump administration has been moving forward with a most-favored-nation pricing plan, which matches the price of a given drug to those set in other major countries. The new pricing will be applied to drugs purchased through the Medicare and Medicaid programs, and Japan is listed as a reference country for price calculations. The pricing scheme under Medicare references prices in 19 countries, including Japan, South Korea, Australia, and various European nations. Manufacturers of the target drugs are required to participate in the Medicare scheme. But drug companies would not need to reference Japan’s drug prices if they do not launch their products there. This raises the possibility of drugmakers refraining from selling products in Japan to avoid lowering prices in the U.S.
STAT+: Google’s clinical director on AI challenges
You’re reading the web edition of STAT’s Health Tech newsletter, our guide to how technology is transforming the life sciences. Sign up to get it delivered in your inbox every Tuesday and Thursday.
Good morning health tech readers!
Does letting general purpose chatbots talk to people with a mental health crisis do more good than harm? Google answers below.
STAT+: Boehringer’s obesity drug shows 16.6% weight loss; more data needed
Want to stay on top of the science and politics driving biotech today? Sign up to get our biotech newsletter in your inbox.
Good morning. I started the morning reading this story of how a botched football kick may have saved someone’s life.
The need-to-know this morning
- Novartis shares were down in early trading after the company reported a drop in sales and operating profit in the first quarter, with generic competition hitting drugs like Entresto hard. The company affirmed its guidance for the year, with net sales expected to grow in the low single digits, and core operating profit forecasted to decline slightly.
- The FDA has taken further actions to withdraw the approval of Amgen’s rare-disease drug Tavneos and remove it from the market. Tavneos was approved in 2021, but new information indicates the drug is ineffective and the application used to support approval contained “untrue statements of material fact,” the FDA said. Amgen has refused to withdraw Tavneos voluntarily, insisting the drug is safe.
Lilly bets on next-gen gene editors with new deal
Eli Lilly this morning announced a deal with AI-focused biotech Profluent to develop new forms of gene editors potentially capable of inserting entire genes into patients.
STAT+: Supreme Court to hear case about ‘skinny labeling’ and generic access
The Supreme Court on Wednesday will hear oral arguments about a simmering controversy over a wonky but important topic called skinny labels, which will be closely watched for its implications for the availability of generic medicines.
Skinny labeling refers to moves by generic companies that seek regulatory approval to market a medicine for a specific use, but not other patented uses for which a brand-name drug is prescribed. For instance, a generic drug could be marketed to treat one type of heart problem but not another. In doing so, the generic company seeks to avoid lawsuits claiming patent infringement.
This tactic has been a key tool for generic companies ever since the Hatch-Waxman Act was signed into law more than four decades ago. The law established the mechanisms by which generic drugs can more readily enter the marketplace. And skinny labeling, which amounts to a carve-out tactic, is one way that Congress attempted to foster more competition and benefit consumers.
Fauci adviser David Morens indicted on charges of concealing emails, avoiding records requests
WASHINGTON — David Morens, a former top National Institute of Allergy and Infectious Diseases official, was indicted Tuesday on allegations that he concealed records from Freedom of Information Act requests.
Trump administration officials said in a legal filing that Morens hid and falsified records to undermine debate about the origins of the virus that spurred the Covid-19 pandemic — and received kickbacks for doing so, including wine and offers of future meals at high-end restaurants. Morens was a senior adviser to former NIAID Director Anthony Fauci, who is not named directly in the indictment.
Opinion: FDA commissioner: ‘Smarter,’ real-time clinical trials could transform drug development
Why does it take a new drug 10 years, on average, to come to market? Part of the reason lies in the dead time in the process.
Historically, trials have required tedious tabulations and repeated application submissions between phases, which is why 45% of the time from a Phase 1 trial until final submission is spent without any ongoing clinical trial in progress — idle time in the system.
Opinion: Healthcare or health care? Help STAT decide
The Associated Press Stylebook, the foundational journalism guide for how newsrooms report and write their stories, made waves last week when it decreed that “health care” should now be written as one word, not two. The change, announced at the annual ACES: The Society for Editing’s conference, was prompted by shifting usage and years of appeals to make the switch.
STAT closely follows AP style and has used “health care” since its founding in 2015. Now, we have to decide whether to adopt this new guidance, but our newsroom is divided.
STAT+: FDA launches effort to speed up clinical trials, using AI
WASHINGTON — The Food and Drug Administration on Tuesday announced efforts to make clinical trials more efficient, starting by reviewing data in real time from trials conducted by AstraZeneca and Amgen.
The agency also asked the public to weigh in on a potential pilot program to work with companies that use AI to enhance safety monitoring and medication dose selections, identify safety signals, and improve patient recruitment in clinical trials.
AstraZeneca is conducting a Phase 2 trial of its combination therapy for patients with an aggressive form of lymphoma. The trial will take place at the University of Texas MD Anderson Cancer Center and the University of Pennsylvania. Amgen is conducting a Phase 1b trial of its treatment for small cell lung carcinoma. The trials will rely on a real-time data platform built by Paradigm Health.
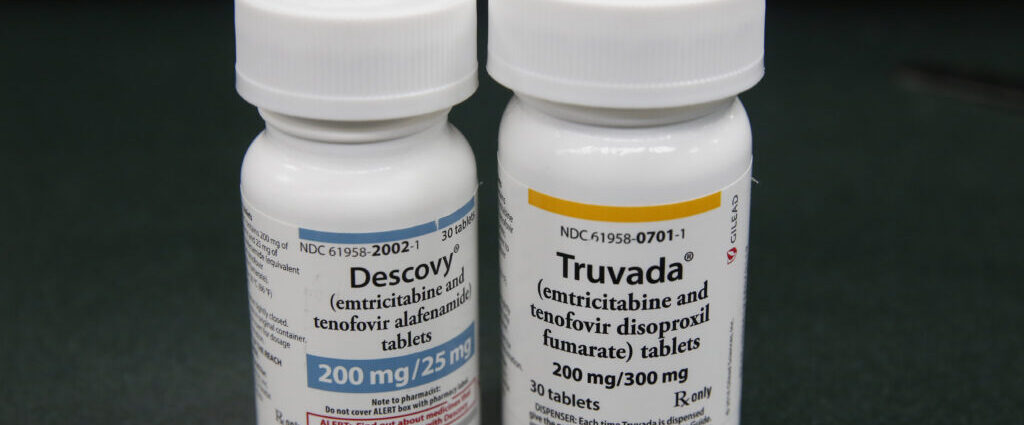
STAT+: AIDS group sues Trump administration over undisclosed agreement with Gilead
An AIDS activist group filed a lawsuit against the Trump administration for failing to disclose a research and development agreement that was at the heart of a settlement between the U.S. government and Gilead Sciences over patents for HIV prevention.
The settlement resolved a contentious lawsuit that was filed six years ago by the previous Trump administration after the Centers for Disease Control and Prevention maintained that Gilead infringed its patent rights. The agency had helped fund academic research that later formed the basis for two Gilead HIV pills, Truvada and Descovy.
The administration had alleged that Gilead ignored the contributions by CDC scientists, exaggerated its own role in developing HIV prevention drugs, and refused to sign a licensing agreement despite “multiple attempts” at reaching a deal after unfairly reaping hundreds of millions of dollars from research funded by taxpayers.
STAT+: Health system CEOs get off easy at Congressional hearing on affordability
The four health system CEOs summoned before a Congressional committee Tuesday likely breathed sighs of relief early in the hearing, when it became clear they had friends in the audience.
Instead, committee members largely blamed the other party’s health care policies for driving U.S. health care prices to levels inaccessible to many Americans.
The hearing was part of the House Ways and Means Committee’s effort to understand the root causes of rising health care costs in the U.S. It comes three months after the committee heard from the CEOs of the country’s largest health insurers, who largely deflected blame onto hospitals and drugmakers.
In attendance were the CEOs of some of the country’s largest health systems: HCA Healthcare, a for-profit system of 190 hospitals, and CommonSpirit Health, a nonprofit system of 158 hospitals. The CEOs of New York-Presbyterian and North Carolina’s ECU Health were also there.
In first meeting, federal autism committee focuses on ‘profound autism’
The federal autism advisory committee met for the first time since Trump took office, months after health secretary Robert F. Kennedy Jr. fired most of the committee’s scientific experts and replaced them with activists and advocates who more closely align with his controversial views on the link between autism, vaccines, and environmental factors.
In its first meeting in 19 months, the Interagency Autism Coordinating Committee, which helps set the federal research agenda and the hundreds of millions that come with it, focused on how to help autistic people with the highest support needs. The group voted on Tuesday to recommend several proposals to help this population, including a directive that would change the existing scientific definition of people with “profound autism.”
New report shows some health equity wins. Experts fear they will be short-lived
A report released Wednesday highlights successes in reducing health disparities in U.S. states. Since the last iteration of the analysis by the Commonwealth Fund, two states expanded eligibility for Medicaid, many states extended postpartum coverage for mothers, and enrollment in Affordable Care Act marketplace plans increased at an unprecedented clip.
But given the report covered the years 2022 to 2024, many equity researchers fear the gains may be short-lived. Outside experts who reviewed the report predict that policies from the second Trump administration, including changes to insurance coverage and vaccine policies and cuts to programs promoting diversity, equity, and inclusion, will exacerbate inequities.
Opinion: STAT+: Did Kennedy just stack the deck on FDA oversight of peptides?
I’ve been waiting for health secretary Robert F. Kennedy Jr. to do something big on oversight of what I call pop peptides, like BPC-157 and GHK-Cu. He had long signaled that he was going to free such peptides from what he saw as a past, misguided FDA that had banned them in 2023.
It’s finally happened — and the way it went down shook me up a bit.
For a few years, a loophole in compounding rules had allowed specialty pharmacies to make and market these peptides. It effectively meant that substances nominated for compounding — even unproven drugs — could be made and marketed by qualified pharmacies while the FDA pondered the nominations. But in 2023, the Food and Drug Administration rightly moved peptides to a no-compounding-allowed status called Category 2 due to concerns about safety and lack of clinical trial data. Now Kennedy is working to undo that with major risks to the public.
Opinion: The medical school nutrition blues
One of health secretary Robert F. Kennedy’s refrains has focused on medical education: Doctors don’t know enough about nutrition and preventive medicine, he likes to say. He has encouraged medical schools to beef up (tallow up?) their education on healthy eating and its connection to chronic disease.
What do medical students think of this?
Opinion: Why alternative medicine can feel so much better than mainstream health care
If all goes well — which is to say if you are generally healthy — you mostly experience the doctor’s office as an inconvenience, going to primary care appointments that are an hour of waiting for 20 minutes of care.
If you’re ill, you fall into a Kafka-esque labyrinth of specialists, waiting rooms, and prior authorizations. You will be held tightly by the medical system, but you are unlikely to feel good.
Mark Cuban Targets Health Insurers, Unveils Consumer-Driven Alternative to Cut Costs
Mark Cuban is intensifying his campaign against the structure of the American healthcare system, arguing that the industry’s profit model is fundamentally misaligned with patient care while advancing a consumer-focused alternative designed to bypass traditional insurers. The billionaire entrepreneur and Cost Plus Drug Company co-founder used a series of public statements and conference remarks to frame the debate not as incremental reform, but as a structural overhaul of how healthcare is financed and delivered in the United States.
In recent posts on X, Mark Cuban sharply criticized major health insurers, arguing they operate less as risk managers and more as financial entities extracting value from system complexity. “Most insurers aren’t insurers,” Cuban wrote, describing large carriers as holding companies that exploit regulatory gaps and contractual inefficiencies across federal, state, and employer-sponsored systems. His comments came in response to healthcare policy expert Larry Levitt of the Kaiser Family Foundation, who questioned what value consumers receive relative to insurer overhead and sustained profitability.
Cuban expanded on these concerns during remarks at the Punchbowl News Conference in Washington, D.C., where he pointed to consolidation across the healthcare ecosystem as a central driver of rising costs. He argued that large insurers’ ownership stakes in pharmacy benefit managers and affiliated services allow them to exert outsized influence over pricing and access. According to Cuban, this structure contributes to high deductibles that leave patients effectively underinsured, forcing hospitals into the role of lenders when individuals cannot meet out-of-pocket obligations.
The critique is paired with a quantitative claim: Cuban has estimated that eliminating insurer-driven administrative complexity and fraud could reduce healthcare costs by 20% to 30%. While that figure remains debated among policy analysts, it underscores his broader argument that inefficiency—not just pricing—is a core driver of U.S. healthcare spending.
To translate that thesis into a practical model, Cuban has outlined a consumer-directed framework informally dubbed “The 10 Plan.” The proposal centers on monthly contributions capped at roughly 10% of household income, designed to mirror the cost of a mid-tier Affordable Care Act plan. Using an example of approximately $2,100 per month for a family of five, Cuban proposes allocating around $300 toward stop-loss insurance with a $30,000 cap, and approximately $200 toward a direct primary care arrangement with a local provider.
The remaining funds—roughly $1,600 in his example—would be directed into a restricted-use medical account, similar to a health savings account but with broader application. Under the model, unused funds would accumulate and earn interest over time, remaining under the control of the account holder until retirement age. For expenses exceeding available balances but falling below catastrophic thresholds, the system would offer a lending mechanism, with future contributions used to repay the borrowed amount.
Cuban’s proposal builds on the transparency-first approach he introduced with the Mark Cuban Cost Plus Drug Company, launched in 2022. That platform bypasses traditional insurance channels, selling medications at cost plus a fixed 15% markup and clearly disclosed fees for pharmacy operations and shipping. By publicly listing drug prices, the company has sought to challenge longstanding opacity in pharmaceutical pricing.
That same transparency principle now extends into his broader healthcare strategy. Through Cost Plus Wellness, Cuban is working to connect self-insured employers directly with healthcare providers via pre-negotiated, publicly available contracts. The platform currently includes dozens of agreements covering thousands of providers and facilities, including participation from Baylor Scott & White Health, one of the largest nonprofit health systems in Texas. Cuban argues that such direct contracting reduces administrative layers while giving employers and patients clearer visibility into pricing.
The timing of Cuban’s renewed push coincides with rising employer healthcare costs, which are projected to increase by more than 9% this year to roughly $17,000 per employee, according to consulting firm Aon. Analysts have pointed to factors such as the rapid adoption of high-cost therapies, including GLP-1 weight-loss drugs, as key contributors to the upward pressure. Against this backdrop, Cuban has called on major insurers to divest non-core assets, arguing that vertical integration has made the market “completely inefficient.”
Cuban has also signaled openness to collaboration with government-led initiatives. He recently commented on the TrumpRx direct-to-consumer drug platform, giving it an “A-” rating as its formulary expanded to include dozens of medications, including fertility treatments and GLP-1 drugs. In remarks to Healthcare Brew, Cuban said, “anything that saves patients money is a good thing,” noting that Cost Plus is working to support broader distribution efforts.
At the center of Cuban’s argument is what he describes as “profit engineering” within the healthcare system—layers of billing practices, coding strategies, and administrative processes that collectively inflate costs. “Healthcare is a simple business that has been made complicated,” he wrote, summarizing his view that systemic complexity, rather than medical necessity, often drives pricing outcomes.
Whether Cuban’s consumer-directed model gains traction will depend on regulatory feasibility, employer adoption, and patient behavior in a system historically anchored in insurance-based risk pooling. Still, his continued push—from pharmaceuticals to primary care financing—signals a sustained effort to reframe healthcare economics around transparency and individual control, challenging one of the largest and most entrenched sectors of the U.S. economy.
JBizNews Desk
Kennedy’s MAHA Agenda Reshapes U.S. Healthcare Economics, From Drug Prices to Hospital Menus
Washington, D.C. — U.S. Health and Human Services Secretary Robert F. Kennedy Jr. is rapidly reshaping the economic foundations of American healthcare, driving sweeping changes that extend far beyond Washington and into pharmaceutical pricing, food production, hospital systems, and rural healthcare infrastructure.
After a series of congressional appearances—including his eighth hearing this month before the House Energy and Commerce Committee—the most consequential developments of Kennedy’s tenure are emerging across the private sector, where policy shifts are translating into measurable cost changes and operational adjustments for major industries.
Fifteen months into leading the nation’s largest civilian agency, Secretary Robert F. Kennedy Jr. has advanced a broad “Make America Healthy Again” (MAHA) agenda that is already producing tangible economic outcomes across pharmaceuticals, agriculture, and healthcare delivery systems.
At the center of the initiative is the administration’s Most Favored Nation (MFN) drug pricing program, implemented through the TrumpRx platform launched in February 2026. The platform initially covered 40 high-cost branded medications, aligning U.S. prices with the lowest levels paid in other developed countries.
The program has since expanded significantly. As of April 23, agreements have been reached with 17 major pharmaceutical companies, including Pfizer, Johnson & Johnson, Merck, Novartis, Bristol Myers Squibb, Amgen, Gilead Sciences, and Regeneron, collectively representing approximately 86% of the U.S. branded drug market. The pricing impact has been substantial: Novo Nordisk’s Wegovy has fallen from roughly $1,350 per month to about $350, while Eli Lilly’s Zepbound has declined from more than $1,000 to approximately $346. Discounts across the platform range from roughly 30% to more than 90% on select therapies.
A recent agreement with Regeneron underscores the program’s expanding scope. The company committed to reducing the price of its cholesterol drug Praluent from $537 to $225 and agreed to apply MFN pricing to all future therapies. The same announcement included FDA approval of a gene therapy for a rare congenital form of deafness, which Regeneron said it would provide at no cost to eligible patients.
Beyond pharmaceuticals, the MAHA initiative is reshaping the food and beverage industry. More than 40% of U.S. food producers have committed to phasing out petroleum-based artificial dyes, while the Food and Drug Administration (FDA) has approved new natural alternatives derived from fruits and vegetables. PepsiCo said it will remove synthetic FD&C colors from several major beverage lines, replacing them with plant-based ingredients.
The initiative is also influencing medical education and clinical practice. More than 50 medical schools have committed to expanding nutrition education from an average of two hours to approximately 40 hours of coursework, reflecting a shift toward prevention-focused care and diet-related health management.
Within hospital systems, Secretary Kennedy directed the Centers for Medicare & Medicaid Services (CMS) to issue updated guidance requiring facilities receiving federal funding to align patient meals with national dietary standards. The directive focuses on reducing ultra-processed foods, sugar-sweetened beverages, and refined carbohydrates—effectively bringing nutrition policy into the core of clinical care.
Rural healthcare has emerged as another key pillar of the agenda. Through the $50 billion Rural Health Transformation Fund, federal officials are channeling resources into underserved communities. This month alone, more than $135 million has been allocated to expand rural residency programs, improve healthcare access, and strengthen nutrition services in remote regions.
Duvi Honig, a national business leader and public policy advocate, praised the Secretary’s leadership and broader impact. “Secretary Bobby Kennedy is making history with a bold, results-driven approach that is transforming healthcare economics in real time,” Honig said. “We are proud to work closely together and support these efforts—from lowering drug prices to advancing critical public health initiatives. We are especially encouraged by the ongoing work with HHS and the CDC on long-overdue Lyme disease awareness and updated clinical guidance, and we look forward to those updates being released.”
On Capitol Hill, Kennedy’s latest testimony also addressed leadership at the Centers for Disease Control and Prevention (CDC), where he voiced support for the nomination of Dr. Erica Schwartz, a former deputy surgeon general with extensive public health experience. Kennedy confirmed he has spoken with Schwartz and supports her candidacy.
As the MAHA agenda continues to take shape, its economic footprint is becoming increasingly visible across multiple sectors. By targeting drug pricing, food standards, hospital practices, and rural health access simultaneously, the administration is advancing a comprehensive approach that is redefining both the cost structure and delivery of healthcare in the United States.
Looking ahead, the durability of these changes will depend on continued industry participation and regulatory execution. But with broad adoption already underway, the MAHA initiative is positioning itself as a defining force in the next phase of American healthcare—where economics, policy, and public health outcomes are more tightly linked than ever.
JBizNews Desk
Supreme Court grapples with multibillion-dollar wave of lawsuits over Roundup cancer claims
WASHINGTON — The Supreme Court seemed divided Monday over whether to block thousands of lawsuits alleging the maker of the weedkiller Roundup failed to warn people it could cause cancer.
The case came before the justices after a tidal wave of litigation that included some multibillion-dollar verdicts against the global agrochemical manufacturer Bayer, which owns Roundup maker Monsanto.
New U.S. recommendation on hepatitis B vaccine will have dire consequences, studies project
A new U.S. policy that recommends offering hepatitis B vaccine at birth only to babies perceived to be at risk of neonatal infection will lead to increased numbers of infected infants and more cases of chronic hepatitis B infection in children that will generate millions of extra dollars in health care costs, two studies published Monday project.
“Avoiding an increase in neonatal infections under the targeted recommendation would require historically unattained levels of maternal [hepatitis B] screening or birth-dose coverage among infants of unscreened mothers,” said one of the studies, from researchers at Boston University, the University of Florida, and Johns Hopkins University.
Trump administration warns against using federal dollars on fentanyl test strips
The Trump administration is doubling down on its opposition to harm reduction services for people who use illicit drugs.
In an open letter on April 24, the federal agency overseeing addiction and mental health policy warned its grantees against using federal funds to buy harm reduction supplies including sterile syringes and pipes, or to distribute test strips for common drug supply adulterants like fentanyl, xylazine, and medetomidine.
STAT+: Dems say RFK Jr. has a pattern of failing to answer their questions
WASHINGTON — Last week, Sen. Ben Ray Luján (D-N.M.) asked health secretary Robert F. Kennedy Jr. whether he would release — by Friday — the contract of a longtime vaccine critic who was hired by the Department of Health and Human Services.
“Yeah, I’m happy to,” Kennedy responded.
But Friday came and went without a response from Kennedy. On Monday, Luján’s office said they plan to follow up with HHS.
STAT+: Erasca touts strong, though preliminary, results in trial of pancreatic and lung cancer therapy
The drugmaker Erasca said Monday that its RAS-targeting pill shrank tumors in 40% of patients with advanced pancreatic cancer and 62% of patients with advanced non-small cell lung cancer, results that the company said exceeded its expectations.
The new data, collected from studies done in the U.S. and China, are still preliminary. However, Erasca said the clinical benefit and tolerability of its drug, called ERAS-0015, compared favorably to daraxonrasib, a similar RAS-targeting drug from Revolution Medicines that recently showed a doubling of overall survival in patients with advanced pancreatic cancer.
“I’m excited about both datasets, but I think lung is more definitive at this point. The pancreatic results are maturing, but are very, very promising,” Erasca CEO Jonathan Lim told STAT. “All options are on the table.”
STAT+: An insider reveals the real goals of prior authorization
This is the online version of STAT’s weekly email newsletter Health Care Inc. Sign up here.
Well, it appears the Associated Press chose violence with this grammar decision. Over my deadbody, AP. This debate has played out so many times, so do I dare ask you, loyal reader: Is it health care or healthcare? bob.herman@statnews.com.
‘The denial is the outcome’
Archelle Georgiou understands Americans’ frustrations with prior authorization. She helped squash the practice before, at what is now the country’s largest health insurance company.
STAT+: Pharmalittle: We’re reading about a Lilly deal, an Intellia CRISPR rare-disease treatment, and more
Good morning, everyone, and welcome to another working week. We hope the weekend respite was relaxing and invigorating because that oh-too-familiar routine of meetings, deadlines, and the like has returned with a vengeance. You knew this would happen, yes? To cope, we are relying, as always, on a cuppa stimulation. Our choice today is ginger honey. Feel free to join us. Remember, no prescription is required. Meanwhile, here are some tidbits to help you along. Best of luck accomplishing your goals today and, of course, do keep in touch. …
Intellia Therapeutics disclosed that a single dose of its gene editing treatment dramatically reduced swelling attacks in patients with a rare genetic disorder in a Phase 3 trial, setting up a potential approval, STAT tells us. The therapy, known as lonvo-z, would be the second approved CRISPR-based medicine, after Vertex Pharmaceutical’s sickle cell treatment Casgevy. Intellia has already initiated a rolling submission with the agency. This would be the first in vivo treatment, meaning it edits patient DNA directly in the body. The question for Intellia and for the field will be how attractive the one-and-done approach will be to patients and doctors. Hereditary angioedema, or HAE, which affects around 20,000 patients in the U.S. and Europe, has seen an influx of new treatments over the last few years.
Sun Pharmaceutical agreed to acquire Organon in a deal valued at $11.75 billion, putting the Indian drugmaker on track to become one of the top three global players in women’s health, The Wall Street Journal notes. Sun, which specializes in generics and is one of the biggest pharma names in India by market cap, will fund the deal through a combination of internal cash resources and financing from banks. Organon, which was formed through a spinoff from Merck, has a portfolio of more than 70 products across women’s health and general medicines, including biosimilars, commercialized across 140 countries. Its 2025 revenue was $6.2 billion. The acquisition will help lift Sun into the top 25 global pharmaceutical companies, with a revenue of $12.4 billion.
STAT+: Astellas retries XLMTM gene therapy after deaths
Want to stay on top of the science and politics driving biotech today? Sign up to get our biotech newsletter in your inbox.
New hair loss data for a sort of amped-up minoxidil is looking very plush. Also Astellas is reentering the clinic with a next-generation gene therapy for XLMTM and Intellia’s CRISPR therapy for hereditary angioedema impresses.
The need-to-know this morning
- Eli Lilly said it is buying Ajax Therapeutics, a privately held developer of blood cancer drugs, for as much as $2.3 billion. Ajax’s lead medicine is a next-generation JAK2 inhibitor currently being evaluated in a Phase 1 study in patients with myelofibrosis.
- Ligand Pharmaceuticals said it is buying Xoma, an aggregator of drug royalties, for $739 million.
- Compass Therapeutics said its drug for advanced biliary cancer delayed tumor progression but failed to prolong survival compared to chemotherapy in a clinical trial.
- BridgeBio shares were higher this morning on reports from the weekend that Pfizer had settled two of three patent cases regarding Vyndamax, its medicine for the heart condition known as ATTR-CM. While terms of the settlement have not been disclosed, any extension of patent protection for Vyndamax is also positive for BridgeBio, which sells a competing ATTR-CM drug called Attruby.
CRISPR therapy slashes rare swelling attacks
Intellia Therapeutics said this morning that its one-time CRISPR treatment lonvo-z cut attack rates by 87% in a Phase 3 trial of hereditary angioedema, STAT’s Jason Mast writes. With more than 60% of patients becoming attack-free and no serious safety signals reported, the therapy is being positioned as a functional cure — or at least a major upgrade over existing options.
What’s the true risk for eating disorders with GLP-1s?
Get your daily dose of health and medicine every weekday with STAT’s free newsletter Morning Rounds. Sign up here.
Good morning. The AP Stylebook broke major news in journalism circles on Friday. Health care, they now say, is one word: healthcare. “Crying, screaming, throwing up,” one STAT staffer said on Slack. “ihateit,” another wrote. We’re clearly still digesting all our feelings, and we’ll let you know soon where we land.
STAT+: Oruka’s long-acting psoriasis therapy posts strong results in mid-stage study
A long-acting injectable treatment for plaque psoriasis helped 63% of patients achieve complete skin clearance in a mid-stage clinical trial, its maker, Oruka Therapeutics, reported Monday.
Separately, Oruka said an updated analysis of blood exposure levels supported the potential for the drug, ORKA-001, to be injected just once per year.
Oruka still needs to conduct late-stage clinical trials, but Monday’s results bolster ORKA-001’s potential to become a new treatment for the autoimmune skin disease with remission rates equal to or greater than current commercial blockbusters Skyrizi and Bimzelx, while requiring fewer injections.
STAT+: Veradermics’ hair loss drug succeeds in late-stage trial
An oral medicine for hair loss successfully spurred hair growth in a late-stage trial, startup Veradermics announced Monday.
Veradermics assessed the pill in two ways: by how many hairs grew within a square centimeter of the scalp, on average, and by how satisfied participants were with the results. Over the course of six months, men who took the drug, known as VDPHL01, either once or twice daily had between 30 and 33 more hairs per square centimeter of scalp. Men in the placebo group grew approximately seven additional hairs.
Between 79% and 86% of men taking VDPHL01 said they saw improvement, along with between 72% and 84% of the clinical trial investigators — results that pleased Reid Waldman, a dermatologist turned Veradermics’ chief executive.
STAT+: Intellia says CRISPR-based treatment for rare disease reduced swelling attacks in pivotal trial
Intellia Therapeutics said Monday that a single dose of its gene editing treatment dramatically reduced swelling attacks in patients with a rare genetic disorder in a Phase 3 trial, setting up a potential approval.
The therapy, known as lonvo-z, would be the second approved CRISPR-based medicine, after Vertex Pharmaceutical’s sickle cell treatment Casgevy. Intellia has already initiated a rolling submission with the agency. It would be the first in vivo treatment, meaning it edits patients’ DNA directly in the body.
In the 80-patient study, volunteers with hereditary angioedema (HAE) who received lonvo-z saw their attack rates drop 87%, relative to placebo. Just over 60% were entirely attack-free over the period, compared to 11% for the placebo patients.
NIH-funded research lags in reporting sex differences, new study finds
Fewer than half of papers published by NIH-funded researchers analyze or report their data by sex, which could make it harder to know what the results mean for men and women, a new study found.
Over a decade ago, the National Institutes of Health set out to promote sex-inclusivity in study design by introducing the expectation that research it funded consider sex as a biological variable (SABV). The guidelines are broad, asking researchers to consider SABV in their design, analysis, and reporting, without mandating that sex differences be examined in the results.
STAT+: Zap in a cap: How one neurotech startup is using a hat to treat depression
Wake up. Brush your teeth. Wash your face.
And put on your lifesaving baseball hat.
That’s right. If you have treatment-resistant depression, this could be the regular morning routine in your future. The hat would activate a blueberry-sized device implanted in your skull that sends a pulse of electricity into your brain.
This is Jacob Robinson’s vision — and it got closer to reality on Friday after the Food and Drug Administration approved a request from Robinson’s startup, Motif Neurotech, to start an initial feasibility trial to test the efficacy of their device in treating depression that hasn’t responded to other treatments. Scientists have been zapping brains to alleviate depression for decades through a method called transcranial magnetic stimulation, or TMS. Motif wants to do the same thing, but with a twist.
What happened to Covid?
In April 2020, people around the globe were struggling to come to grips with the strictures of unprecedented societal shutdowns aimed at slowing the spread of Covid-19. Flattening the curve, in 2020-speak.
Six years later, school and business closures, mask wearing, and social distancing are dim, unpleasant memories. And Covid, though it still animates political animus plenty, feels like a threat from yesteryear.
Opinion: Patients seeking mental health treatment are not commodities
As I read over an employment contract for a job as a psychiatric nurse practitioner, one clause stopped me cold. If I left the practice, I’d owe $7,500 for every patient who chose to continue treatment with me.
When I questioned the clause, the response came quickly, with irritation: “The practice owns the patients. You do not.”
Opinion: I’m a fourth-year med student, but I only learned one historical example of medical racism
Last December, news broke that the Centers for Disease Control and Prevention had awarded $1.6 million to a Danish vaccine research group to study the effects of the hepatitis B vaccine on infants in the West African nation of Guinea-Bissau. The proposed five-year study compared outcomes between infants vaccinated at birth and those vaccinated at six weeks of age. As a medical student and researcher, I was shocked by the brazen inequity of the trial.
Randomized controlled trials have already demonstrated superior outcomes when the hepatitis B vaccine is administered at birth. More troubling, however, was the setting of the study. Guinea-Bissau is a highly endemic country for hepatitis B, where vaccination coverage lags behind global averages and roughly 60 percent of the population lives in poverty.
STAT+: Doctor, wife of acting U.S. attorney general, appointed to NIH advisory council
Kristine Blanche, an integrative medicine doctor and wife of acting Attorney General Todd Blanche, has been named as a member to one of the advisory councils that provides critical funding recommendations to the National Institutes of Health. Her appointment, to serve on the advisory council to the National Center for Complementary and Integrative Health, is the first of such appointments to be made in over a year.
It’s unclear if Blanche’s selection — which has not been publicized by the NIH — is a sign of a thawing in the pipeline of advisory council appointments. But it’s done little to quiet simmering concerns among the wider research community about whether the Trump administration would attempt to stack councils with ideological allies who will use their positions to advance its political goals.
It’s “the worst kind of political patronage,” Joshua Gordon, a former director of the National Institute of Mental Health, told STAT. He and others worry the move will erode taxpayers’ trust in how the largest funder of biomedical research in the world spends its $48 billion budget. “It’s clearly meant to contribute to an intentional degradation of confidence in the NIH.”
Nancy Cox, a CDC veteran and a stalwart in global flu research, dies at 77
Nancy Cox, who for decades was a global leader in influenza research, has died. Cox headed the influenza team at the Centers for Disease Control and Prevention for 22 years, shepherding it from a branch of 14 people to a division of over 100. She was also director of the World Health Organization’s Collaborating Center for the Surveillance, Epidemiology, and Control of Influenza at the CDC.
Cox died Thursday from glioblastoma, a cancer of the brain. She was 77.
Opinion: The podcast telling the stories behind Ambien, Ozempic, EpiPens, and other game-changing drugs
Below is a lightly edited, AI-generated transcript of the “First Opinion Podcast” interview with Thomas Goetz. Be sure to sign up for the weekly “First Opinion Podcast” on Apple Podcasts, Spotify, or wherever you get your podcasts. Get alerts about each new episode by signing up for the “First Opinion Podcast” newsletter. And don’t forget to sign up for the First Opinion newsletter, delivered every Sunday.
Torie Bosch: Whether it’s Ambien or Wegovy, ivermectin or fluoride, every drug in your medicine cabinet or advertised on TV has a story behind it. Not just how it came to be, but how it ends up affecting society in unexpected ways, big or small.
STAT+: FDA to speed up review of three psychedelics as mental health treatments
The Food and Drug Administration will accelerate its review of psychedelic drugs developed by Compass Pathways, the Usona Institute, and Transcend Therapeutics for mental health disorders, as part of the Trump administration’s plan to boost access to the controversial yet promising medications.
The agency will grant priority review vouchers specifically to Compass’ psilocybin product for treatment-resistant depression, Usona’s similar medicine for major depressive disorder, and an MDMA-like treatment for post-traumatic stress disorder from Transcend.
The FDA identified the medications receiving the vouchers, but not the companies developing them. Compass, Usona, and Transcend confirmed they received vouchers.
STAT+: Utah medical board calls for immediate suspension of state’s AI doctor experiment
Utah’s high-profile experiment with using an artificial intelligence system to renew prescriptions without physician oversight is facing its first major challenge as doctors in the state push back.
In a letter published Friday, Utah Medical Licensing Board said it only learned about the pilot program with AI doctor startup Doctronic after it had been launched, and asked the state to halt the program.
“Proceeding with this agreement without consulting the Medical Board potentially places Utah citizens at risk and remains a major concern of the board,” they wrote. “It is the strong recommendation of the Utah Medical Licensing Board that this program be immediately suspended pending further discussion.”
STAT+: A biotech VC on what Eli Lilly saw in a struggling cancer startup for $3.2B
Kelonia Therapeutics became the newest biotech takeout target this week. The privately held company, which is developing cell therapies for cancer and autoimmune diseases, will be acquired by Eli Lilly.
The acquisition is a boon for the small startup, which has subsisted on $60 million over the last five years and previously struggled to stay afloat. (Check out an earlier slide deck and memo on the company here.) Kelonia came within a week of running out of cash three times. Now it’s being bought for $3.2 billion with potential milestone payments that could double that payout.
On this week’s edition of its biotech podcast, “The Readout Loud,” STAT spoke with Bryan Roberts, a partner at VC firm Venrock, which incubated the biotech, to discuss how this small company managed to land a big deal.
International childhood vaccine campaign grows
Get your daily dose of health and medicine every weekday with STAT’s free newsletter Morning Rounds. Sign up here.
It’s my birthday tomorrow.
STAT+: Sanofi MS drug gets positive review in Europe following FDA
Regeneron is in the headlines again: It cut a pricing deal with the Trump administration and won approval for a hearing loss gene therapy. Also, Medicare and the FDA are getting creative with their efforts to speed medical device coverage.
And since it’s totally worth it, sign up for STAT+ between now and Saturday and you’ll get 60% off your first year.
MS drug rejected by FDA gets warmer reception in Europe
From STAT’s Andrew Joseph: A multiple sclerosis drug from Sanofi that was rejected late last year by the FDA found a warmer audience in European regulators, who recommended today that the medicine should be approved.
STAT+: Pharmalittle: We’re reading about a Trump deal with Regeneron, reclassifying medical marijuana, and more
And so, another working week will soon draw to a close. Not a moment too soon, yes? This is, you may recall, our treasured signal to daydream about weekend plans. Our agenda is still shaping up but we expect to drop in at a couple of folkie happenings, promenade with the official mascots, and tidy up around the castle. We also hope to have yet another listening party, where the rotation will likely include this, this, this, this and this. And what about you? This is a fine time to enjoy the great outdoors. Hiking trials, shorelines, and cityscapes await you. This may also be an opportunity to start your spring cleaning, read a good book, or search out someone special. Well, whatever you do, have a grand time. But be safe. Enjoy, and see you soon. …
President Trump heralded a drug-pricing agreement with Regeneron Pharmaceuticals, closing the last of 17 deals initially sought by the White House last year, STAT notes. Regeneron, as part of the private deal, will reduce prices on drugs to Medicaid, provide the Praluent cholesterol medicine on TrumpRx for $225, and invest $27 billion in drug development in the United States. At the same time, Regeneron also announced the U.S. Food and Drug Administration approval of Otarmeni, the first gene therapy to be greenlit under the agency’s new National Priority Voucher program. Last year, Trump sent letters to 17 of the largest pharmaceutical companies, demanding lower prices and kicking off months of intense negotiations. But the deals the administration struck with each firm remain private, and in some cases they may not be finalized.
The Trump administration issued an order reclassifying state-licensed medical marijuana as a less-dangerous drug, a major policy shift long sought by advocates who said cannabis should never have been treated like heroin by the federal government, the Associated Press explains. The order signed by acting Attorney General Todd Blanche does not legalize marijuana for medical or recreational use under federal law. But it does change the way it is regulated, shifting licensed medical marijuana from Schedule I, which is reserved for drugs without medical use and with high potential for abuse, to the less strictly regulated Schedule III. The move — which STAT described as “a new era in GOP drug policy” — also gives licensed medical marijuana operators a major tax break and eases some barriers to researching cannabis.
Growing use of guest editors has turned some journals into a ‘playground of bad science’
Should academic journals begin to second guess guest editors?
That question gained new urgency last week when the British Medical Journal’s publishing group retracted nearly its entire guest-edited special edition of the Journal of Medical Genetics, dedicated to cancer immunotherapies. In the retraction note, the journal writes that it was, in part, because of “compromised peer review in almost all articles.” The notice garnered attention for its scope, but also because it exemplified larger concerns that research integrity advocates have with guest-edited editions, which are also called special issues in some journals.
Opinion: The local news crisis is also a public health crisis
The past four months have been a whirlwind for Pittsburgh’s journalism landscape. On Jan. 7, the Pittsburgh Post-Gazette, Western Pennsylvania’s largest news organization, announced it would cease publication on May 3 after nearly 240 years. Then, on April 14, just over two weeks before that closure date, the Baltimore-based Venetoulis Institute for Local Journalism said it would acquire the paper’s assets and continue publication.
Like many Pittsburghers, I experienced the emotional rollercoaster of anger, disappointment, hope, and relief tied to these announcements. I grew up in the Pittsburgh area, where I vividly remember running barefoot down my driveway as a child to grab the Post-Gazette. Years later, I interned there as a health and science reporter and have since contributed as a freelancer.
Psychedelics get a boost from the White House
President Trump recently signed an executive order which aims to increase access to psychedelic drug treatments. He was joined at the signing by podcaster Joe Rogan, who said he’ ha’d messaged the president about research on the psychedelic ibogaine.
In this week’s STATus Report, host Alex Hogan chats with STAT Washington correspondent Daniel Payne about what the executive order does and doesn’t do. Hogan also looks at why ibogaine, and psychedelic drugs more broadly, are increasingly being taken seriously for stubbornly hard-to-treat conditions like addiction, depression, and PTSD.
Opinion: I started medical school at 69 and will begin residency at 72. Here’s what I learned
Since I was 7, my goal has been to become a doctor. But life had other plans. I grew up in a blue-collar family in Levittown, N.Y., in the 1950s and ’60s, so it often felt like the world ended in Jersey. When I landed in Lansing, Mich., to attend Michigan State, I expected the Rocky Mountains to be visible. I ended up getting a degree in nursing, but I always had another goal: to become an M.D.
This year, at the age of nearly 73, my dream will finally come true. Soon after, I will start my residency in family medicine. My perspective on medical school and medicine is unique not only because I attended late in life, but because it came after more than 40 years as a nurse practitioner.
A CAR-T biotech’s dramatic turnaround, and drugmakers’ tactics to drive more scripts
How did a biotech company that almost ran out of money three times get acquired for over $3 billion? Will the M&A streak continue? And why are drugmakers working with a telehealth company called Prescribery?
We discuss all that and more on this week’s episode of “The Readout LOUD,” STAT’s weekly biotech podcast. Venture capitalist Bryan Roberts joins us to discuss his firm’s investment in Kelonia Therapeutics, the CAR-T biotech that Eli Lilly just said it would buy for $3.25 billion.
STAT+: Trump’s boosting of psychedelics, cannabis signal a new era in GOP drug policy
The days of “Just Say No,” it seems, are long gone.
Over the weekend, President Trump signed an executive order to increase the availability of certain psychedelics as treatments for mental health conditions, ordering that $50 million be spent, and that the Food and Drug Administration fast-track reviews to usher in their approval. At one point, the president joked to the motley assembly of administration officials, a former Navy SEAL, and the podcaster Joe Rogan: “Can I have some, please?”
On Wednesday, the Trump administration announced it had downgraded medical marijuana from the highest tier of controlled substances, and was pushing the Drug Enforcement Administration to do the same for recreational marijuana.
The president’s lenient tack on some mind-altering drugs ushers in a new world of right-wing drug policy. While the administration has emphasized hardline, militaristic tactics when it comes to fentanyl, its recent actions on “softer” drugs could represent a new era not just for Republican politics but also for American drug policy writ large.
“With this imminent move, we are now confronted with the most pro-drug administration in our history,” Kevin Sabet, the CEO of the anti-legalization advocacy group Smart Approaches to Marijuana, said in a statement. “Policy is now being dictated by marijuana CEOs, psychedelics investors, and podcasters in active addiction — it is a travesty and injustice to the American people of unprecedented proportions. The marijuana industry is the new Big Tobacco, and President Trump is welcoming them to the homes of families across this country with open arms.”
Heart’s beat may help it beat cancer, mouse research suggests
Heart disease and cancer are the leading causes of death in the United States, but it is rare that cancer makes its way to the heart.
It’s an observation that clinicians have been grateful for, though largely unable to explain. But in a paper published Thursday in Science, researchers propose one potential explanation: The constant pressure that the organ is under from beating thousands of times a day and pushing gallons of blood creates an environment that is hostile to cancers. The study, which was conducted in mice, is preliminary, but outside experts said it points to potential new approaches for cancer treatments.
STAT+: Trump celebrates closing first round of drug pricing deals, promises more ahead
WASHINGTON — President Trump heralded a drug pricing agreement with Regeneron on Thursday, closing the last of 17 deals initially sought by the White House last year.
Regeneron, as part of the private deal, will reduce prices on drugs to Medicaid, provide cholesterol medicine Praluent on TrumpRx for $225, and invest $27 billion in drug development in the United States.
On the same day, Regeneron also announced Food and Drug Administration approval of Otarmeni, the first gene therapy to be greenlit under the agency’s new National Priority Voucher program. In early trials, the drug provided modest hearing gains for people with a rare type of hearing loss, though its development has received pushback from parts of the Deaf community. Regeneron plans to offer the drug at no cost to American patients.
STAT+: Pharmalittle: We’re reading about cheap telehealth visits, pharma withholding meds in Europe, and more
Rise and shine, everyone, another busy day is on the way. Already, the Pharmalot campus has been bustling as the official mascots have been busy chasing assorted creatures in search of their breakfast. As for us, yes, we are firing up the trusty kettle once again and brewing another cuppa stimulation. Our choice today is Earl Gray. Now, though, the time has come to get still busier. On that note, here are some tidbits. Have a lovely day and do keep in touch. …
As links between pharmaceutical companies and telehealth providers grow, health policy experts and legislators are raising concerns over the large fees that telehealth companies can receive from drugmakers each year, STAT explains. Critics have questioned whether those partnerships break federal laws prohibiting financial kickbacks to induce prescribing, highlighting their potential to promote uncoordinated care and overprescriping of unnecessary, and often expensive, branded medications. The same questions apply to coupons extended for a drug-specific telehealth visit. Drugmakers have long used discount coupons to encourage patients to use their high-cost medications. But coupons can influence not just the out-of-pocket price for the drug, but the cost of consulting with a clinician who can prescribe it.
U.S. Health and Human Services Secretary Robert F. Kennedy Jr. told a Senate hearing on Wednesday that he had nothing to do with a U.S. Food and Drug Administration decision to not approve Replimune’s advanced skin cancer drug, saying it was in the hands of FDA Commissioner Marty Makary, Reuters writes. Earlier this month, the FDA declined to approve the drug after taking issue with the company’s reliance on a single‑arm study for the drug without a control group. In its letter, the FDA said the company must provide data from a well-controlled trial demonstrating adequate evidence of effectiveness. “This decision comes out of FDA, and we trust the process there. And I’ve been told by Marty Makary that every panel that looked at that drug unanimously voted against it … because it does not appear to work,” Kennedy said. An op-ed in The Wall Street Journal maintained Kennedy’s comments were not true and cited cancer doctors who have worked on trials of the drug and said it was effective.
Trump reclassifies state-licensed medical marijuana as a less-dangerous drug in a historic shift
WASHINGTON — President Donald Trump’s acting attorney general on Thursday signed an order reclassifying state-licensed medical marijuana as a less-dangerous drug, a major policy shift long sought by advocates who said cannabis should never have been treated like heroin by the federal government.
The order signed by Todd Blanche does not legalize marijuana for medical or recreational use under federal law. But it does change the way it’s regulated, shifting licensed medical marijuana from Schedule I — reserved for drugs without medical use and with high potential for abuse — to the less strictly regulated Schedule III. It also gives licensed medical marijuana operators a major tax break and eases some barriers to researching cannabis.
STAT+: AMA calls for regulatory crackdown on chatbots, wellness AI
You’re reading the web edition of STAT’s Health Tech newsletter, our guide to how technology is transforming the life sciences. Sign up to get it delivered in your inbox every Tuesday and Thursday.
Good morning health tech readers!
After inviting consumers to upload medical records for examination by ChatGPT earlier this year, OpenAI is now making its play for their doctors as well.
STAT+: RFK Jr. backs off some MAHA issues
You’re reading the web edition of D.C. Diagnosis, STAT’s twice-weekly newsletter about the politics and policy of health and medicine. Sign up here to receive it in your inbox on Tuesdays and Thursdays.
Sarah Todd’s MAHA snackdown article includes such merchandising gems as “fibermaxxing” chips, “protein-powered” yogurt, and “longevity” beverages. My generation’s biggest contribution to food marketing was light beer. Send news tips and brand management schemes to John.Wilkerson@statnews.com or John_Wilkerson.07 on Signal.
Losing BALANCE
The Trump administration couldn’t get insurers to provide cheap obesity drugs to seniors through the Medicare pilot BALANCE. So Medicare will now make the drugs available to seniors outside of the Part D drug benefit in another pilot, called Bridge.
STAT+: Legislatures in Colorado and Virginia resist moves to constrain drug affordability boards
Legislators in two states have resisted efforts to restrict Prescription Drug Affordability Boards, the controversial panels that are designed to function as rate-setting authorities and place limits on the cost of prescription medicines.
In Virginia, the General Assembly unanimously rejected a move by Gov. Abigail Spanberger (D) to delay a key provision of two bills that would create a board and allow it to place price caps that mirror the negotiated prices paid by Medicare. Spanberger must now either accept or veto the legislation as originally intended.
In Colorado, the House Health and Human Services Committee postponed consideration of a bill that would exempt orphan drugs, which are used to treat rare diseases, from pricing caps that might be pursued by the state board. By delaying action until the end of the legislative session, the bill has effectively died.
STAT+: CMS, FDA team up to fast-track reimbursement for breakthrough devices
Federal regulators on Thursday announced a new Medicare coverage pathway for devices deemed breakthroughs by the Food and Drug Administration. The program aims to address longstanding industry complaints that it takes too long for Medicare to pay for innovative technology.
The new Regulatory Alignment for Predictable and Immediate Device coverage pathway, or RAPID, will try to synchronize the FDA’s premarket review process, which determines whether products are safe and effective, and Medicare’s independent process which determines whether paying for devices is reasonable and necessary. The goal is to collect and review enough data before authorization that Medicare can cover the products soon after a product hits the market. It’s the latest effort to speed up patient access to breakthrough devices.
“We’re going to demystify the process by providing a clear signal to device manufacturers of exactly what targets they need to hit to get Medicare coverage,” said Medicare Deputy Administrator John Brooks. “And that’s enormously valuable, if you think about the investment timelines for device manufacturers who are trying to bring life-saving treatments to individuals.”
STAT+: Sanofi research priorities in flux as new CEO logs in
Want to stay on top of the science and politics driving biotech today? Sign up to get our biotech newsletter in your inbox.
A gene therapy for deafness is starting to deliver real-world benefit. Also, ultra-cheap telehealth options are raising concerns about whether easier access to prescription drugs is coming at the expense of patient care. And, in front of Congress, RFK Jr. seems to be striking a different tune on vaccines.
Sanofi research priorities in flux as new CEO logs in
From my colleague Andrew Joseph: One of the open questions about the future of Sanofi as Belén Garijo takes over as CEO this month is whether the company, which had upped its focus in immunology in recent years only to come up with lackluster results, will rebalance its priorities.
Opinion: Dropping the flu vaccine requirement puts U.S. military readiness at risk
On Tuesday, Secretary of Defense Pete Hegseth announced that the men and women who defend our nation will be able to choose whether to be protected from the flu. In his video statement, he said that service members will no longer be required to get the annual influenza vaccine because flu causes “no threat to our military readiness.”
Unfortunately, this is untrue. During the 1918 influenza pandemic, more than 20,000 U.S. service members died and hundreds of thousands were hospitalized. People of military age were among those with the highest death rates during that outbreak, impacting not only their health, but the U.S. economy and military readiness. Historians report that up to 20% of U.S. service members became ill. As a result, the U.S. military began pursuing a flu vaccine to ensure that, in the future, our nation’s defenders would be protected from this threat.
Across 7 hearings, RFK Jr. talks a new, more moderate MAHA
Get your daily dose of health and medicine every weekday with STAT’s free newsletter Morning Rounds. Sign up here.
Good morning, we made it to Thursday. Thanks for reading.
STAT+: Can Erasca be biotech’s next big thing? We’ll see
This is the online version of Adam’s Biotech Scorecard, a subscriber-only newsletter. STAT+ subscribers can sign up here to get it delivered to their inbox.
I wore my Tottenham Hotspur hoodie while shopping at Market Basket last Sunday. One fellow shopper laughed at me. He must have been an Arsenal fan. But another guy commiserated.
If none of this means anything to you, I’m sorry. My favorite soccer team is circling the drain and I feel sad.
The promise of a better pan-RAS inhibitor
Erasca has been described as the poor man’s Revolution Medicines. Impoverished doesn’t exactly fit, not with Erasca’s market value nearing $7 billion on the promise of a better pan-RAS inhibitor for pancreatic cancer. But RevMed’s value now tops $30 billion, so you can see why biotech investors are motivated to find the next big thing.
Whether Erasca is worthy of that description will become clearer in May when the company reports initial results from an early stage study of its drug, called ERAS-0015.
“RevMed has been a real pioneer in this space,” Erasca co-founder and CEO Jonathan Lim told me when we spoke on Tuesday. “What a day it was last week seeing their data with 13.2 months of median overall survival. It’s great for patients with pancreatic cancer.”
Opinion: Kennedy’s new silence on vaccines is political — and it won’t last
For the moment, Health and Human Services Secretary Robert F. Kennedy Jr. seems quieter and more positive on vaccines than many, us included, ever expected he could be. Public messaging has shifted — at least superficially — toward nutrition, chronic disease, and the Make America Healthy Again agenda. Kennedy even acknowledged in a recent congressional hearing that the measles, mumps, and rubella vaccine is safe and effective “for most people.”
But this lull in anti-vaccine rhetoric and action should not be mistaken for a durable pivot in federal vaccine policy. It is a cynical, political pause: the eye of a storm shaped more by electoral timing than public health strategy.
STAT+: Will bargain-basement telehealth visits help pharma drive drug scripts?
The little pink pill recently got a retail boost. Addyi, a drug that treats low libido in premenopausal women, has been on the market for more than a decade after a controversial approval. But in December the Food and Drug Administration expanded its use, approving the daily drug for all women under 65.
More women than ever now could qualify for the pill — and its maker, Sprout Pharmaceuticals, made it easy for them to find out. On its Instagram account, in Facebook ads, and on its website, Sprout shared how women could get a quick consult for an Addyi prescription: With a special code, PINKPILL, they could get a telehealth visit for just $10.
Drugmakers have long used discount coupons to encourage patients to use their high-cost medications. Now, coupons can influence not just a drug’s out-of-pocket price, but the cost of consulting with a clinician who can prescribe it.
For Addyi, those consultations and discounts are managed by a telehealth company called Prescribery — one of several that work with drugmakers to help patients “talk to a doctor now” about their products. “We give them the coupon codes that they can use, and they get to market it to drive additional business,” said Prescribery’s CEO and CFO Ross Pope. “That’s sort of our arrangement, where they’re driving more business, both for them and for us.”
As links between pharmaceutical companies and telehealth providers grow, health policy experts and legislators have sounded alarms. Telehealth companies can receive hundreds of thousands of dollars in pharma fees a year. Critics have questioned whether those partnerships break federal laws prohibiting financial kickbacks to induce prescribing, highlighting their potential to promote uncoordinated care and overprescription of unnecessary, and often expensive, branded medications.
The same questions, they say, apply to coupons extended for a drug-specific telehealth visit. “These discount structures are one more piece of the same puzzle,” said Daniel Eisenkraft Klein, a postdoctoral fellow at the Program on Regulation, Therapeutics, and Law at Brigham and Women’s Hospital and Harvard Medical School. “It’s this big financial architecture that’s every step of the way designed to move patients toward the specific drug cheaply and quickly.”
STAT+: Publicly, Kennedy embraces a more moderate MAHA
WASHINGTON — Health secretary Robert F. Kennedy Jr. once said there are no vaccines that are safe and effective. On Wednesday, he seemed to have changed his tune.
Across two Senate hearings, Kennedy noted that as health secretary, he funded the development of new vaccines, green-lit new shots for patients, asserted flu vaccines are preventive care, and even urged “every child to get the MMR,” a shot he previously suggested wasn’t safe. Last week, he acknowledged the shot could have saved the life of a child who died of measles.
Kennedy’s agenda continues to make waves across American health care, as his department pursues a broad crackdown on alleged fraud and seeks to upend Americans’ relationship with ultra-processed foods, all after major cuts across health agencies and a reworking of vaccine policy. But the about-face expands to a number of core MAHA issues — chemicals in food and the government’s relationship with industry among them.
It comes as some leaders of the insurgent movement have grown skeptical of the administration they rallied to support, forcing the Trump administration to thread the needle between courting the MAHA base’s ongoing support and dropping MAHA priorities seen as impractical or politically unwise.
Former tobacco executive joins CDC senior leadership, raising concerns over industry influence
A former tobacco industry executive has been appointed to senior leadership at the Centers for Disease Control and Prevention, alarming public health advocates and critics of industry influence on government.
Stephen Sayle, named in March as the CDC’s deputy director for legislative affairs, previously worked at Fontem Ventures, a subsidiary of the British multinational tobacco corporation Imperial Brands. Between 2017 and 2018, he was U.S. vice president of corporate affairs at Fontem, which is focused on non-combustible tobacco products like the e-cigarette brand blu and the oral nicotine pouch brand Zone.
STAT+: Federal test of AI prior authorization is delaying care for seniors, report says
Washington state hospitals say their Medicare patients are waiting two to four times longer in some cases for procedures that are now subject to prior authorization under a new Medicare program.
The report from U.S. Sen. Maria Cantwell (D-Wash.) is among the first to document alleged patient harm stemming from the Centers for Medicare and Medicaid Services’ new Wasteful and Inappropriate Service Reduction, or WISeR, Model. Cantwell is one of several Democratic members of Congress who have been urging CMS to scrap the program, which launched Jan. 1.
Cantwell aired her concerns about WISeR to Health and Human Services Secretary Robert F. Kennedy Jr. at a Senate Finance Committee hearing on Wednesday. She said CMS is using AI as a “denial device” and that patients are waiting weeks to get approval for services that previously didn’t require approval.
Video: Robert F. Kennedy Jr. testifies before Senate HELP committee
Health secretary Robert F. Kennedy Jr. testified on Wednesday before the Senate lawmakers who arguably hold the most power in advancing or hindering his Make America Healthy Again agenda.
The Senate Health, Education, Labor, and Pensions Committee hearing put the secretary face-to-face with Chair Bill Cassidy (R-La.), whose vote to confirm Kennedy last year came with a number of promises on vaccine policy that Kennedy has since blown through.
STAT+: At AACR, talk of Chinese biotech, oncology’s comms issue, and more
You’re reading the web version of STAT’s popup newsletter, AACR in 30 seconds, your guide to what’s happening at the American Association of Cancer Researchers’ annual meeting.
This is the last edition of our pop-up newsletter. We hope you’ve learned as much as we have. If you’re not already a STAT+ subscriber, consider it! There’s currently a 60% off promotion on annual subscriptions.
In the meantime, thanks for joining us.
Overcoming resistance and RevMed’s next drug?
In case you missed it, Revolution Medicines’ sessions yesterday were jam-packed with conference attendees. While most of the media coverage focused on the daraxonrasib in frontline pancreatic cancer data, the company also revealed some activity in a new compound, RM-055. CEO Mark Goldsmith described it as being part of a new class of “catalytic inhibitors,” since it can slice off a phosphate from GTP-RAS, or the “on” form of RAS, and turn the protein off.
This generated a lot of interest because one of the main ways that cancer develops resistance to RAS inhibitors is by amplifying mutant RAS, basically flooding the cell with the oncoprotein and overwhelming the inhibitor. RM-055, with its catalytic ability to turn multiple mutant RAS proteins off, may be the next step in the arms race against RAS-addicted cancer.
Live: Robert F. Kennedy Jr. testifies before Senate HELP committee
Health secretary Robert F. Kennedy Jr. will testify Wednesday before the Senate lawmakers who arguably hold the most power in advancing or hindering his Make America Healthy Again agenda.
The Senate Health, Education, Labor, and Pensions Committee hearing will also put the secretary face-to-face with Chair Bill Cassidy (R-La.), whose vote to confirm Kennedy last year came with a number of promises on vaccine policy that Kennedy has since blown through.
STAT+: The latest biotech news: RevMed, Roche, Kyverna CAR-T therapy
Want to stay on top of the science and politics driving biotech today? Sign up to get our biotech newsletter in your inbox.
Good morning. We’ve got a lot of new data to dig into, so let’s get straight to it.
Medicare delays pilot for covering obesity drugs
One of the key health care accomplishments the Trump administration wanted to claim was securing coverage of obesity drugs for seniors. That plan is now unraveling.
Increased calls to 988 hotline associated with a drop in suicides among young people, study finds
After the U.S. launched 988, a new shortened phone number for the national suicide and crisis hotline, suicide deaths among adolescents and young adults dropped 11% compared to projected rates, according to a JAMA study published Wednesday.
Suicide rates have been increasing in the U.S. for decades, especially among young people. Overall, the number of suicide deaths in the U.S. dropped slightly in 2024 from a peak in 2022 — the year that 988 was launched. The new research found even greater improvements when comparing the quarterly rate of suicide deaths among people age 15 to 34 against projections. And the states that saw the biggest increases in answered calls to the lifeline experienced the largest decline in suicide rates.
STAT+: Gene therapy trial for deafness adds evidence to drug’s efficacy
Three years after researchers first injected deaf children with a treatment designed to give them hearing, an early scientific consensus is emerging: This gene therapy works.
A team of researchers published on Wednesday the most promising results to date: In a clinical trial in China, 90% of participants reported significant improvement in their hearing after receiving an injection. The therapy even improved hearing in a few adults, including a 32-year-old. The results were published in Nature.
“In some patients, the hearing improved so well that they can hear whispers,” said Zheng-Yi Chen, a study author and Mass Eye and Ear otolaryngologist. “To reach a stage where you can hear normal conversation, it’s mind-boggling.”
STAT+: Pharmalittle: We’re reading about Medicare’s obesity drug pilot, an experimental Roche MS drug, and more
Good morning, everyone, and welcome to the middle of the week. Congratulations on making it this far, and remember there are only a few more days until the weekend arrives. So keep plugging away. After all, what are the alternatives? While you ponder the possibilities, we invite you to join us for a delightful cup of stimulation. Our choice today is maple cinnamon French toast. Remember that no prescription is required — so no co-pay or rebate is involved. Meanwhile, here is the latest menu of tidbits to help you on your way. Have a wonderful day and please do stay in touch. …
The Trump administration had planned for insurers to provide cheap weight loss drugs to seniors through a Medicare pilot program, but insurers said no, so the government will instead cover the drugs outside of the Medicare Part D drug benefit, STAT explains. Last year, the Trump administration struck a deal with Eli Lilly and Novo Nordisk to lower the prices of their popular obesity drugs in exchange for expanding access to those drugs in Medicare and Medicaid. The hitch was that insurers had to agree to participate in the pilot, called BALANCE, even though they would likely have faced a financial strain by doing so. The drugmakers agreed to sell the treatments for $245 a month in Medicare and Medicaid, but Medicare promised that beneficiaries would only pay $50 each month. And the pilot could only proceed if Medicare Part D drug plans covering at least 80% of enrollees agreed to participate.
Amazon is launching a program that provides access to GLP-1 treatments through its One Medical primary care arm, The Wall Street Journal notes. The program is designed to provide continuing medical supervision — allowing clinicians to monitor progress, adjust treatments, and address related health conditions — compared with one-off weight-management solutions from other providers. With the program, customers can get Novo Nordisk’s Wegovy and Eli Lilly’s Foundayo weight loss pills starting at $25 a month with insurance coverage, or $149 a month for cash-pay options. The cash-pay price for a starter dose aligns with those offered through programs from Hims & Hers Health, Walgreens, and Weight Watchers. Several companies in the weight loss market saw their stocks pull back after Amazon’s announcement. Shares of Hims & Hers closed down 4% at $29.76 on Tuesday. Weight Watchers declined 8.8%, and Novo Nordisk fell 2.6%.
Moderna continues bird flu vax study, but limits work in the U.S.
Get your daily dose of health and medicine every weekday with STAT’s free newsletter Morning Rounds. Sign up here.
Good morning. Big news: I’ve convinced at least one other STAT staffer to re-read “The Odyssey” with me ahead of the movie this summer. Starting today, that means we’ll read three books (chapters), or about 1,500 lines, per week for the next eight weeks. Care to join us?
Opinion: Behind every miracle drug is a story
Have you ever wondered how many drugs are behind the counter at your local pharmacy?
According to Thomas Goetz — a journalist, entrepreneur, and host of the new podcast “Drug Story” — “there are over 3,000 drugs behind a typical pharmacist counter.” And behind each drug is a story.
Are MAHA snacks really better for you? Nutrition experts parse a grocery aisle gold rush
Vani Hari has 2.3 million followers on Instagram, and about as many ideas for healthy food swaps. An entrepreneur and influential food activist in the Make America Healthy Again movement, Hari gives regular shout-outs to substitutes for snacks that contain corn syrup, seed oils, and other ingredients on health-conscious Americans’ blacklist.
For Valentine’s Day, YumEarth choco yums instead of artificially dyed M&Ms. (“Let me say these treats are BETTER, but they are still candy,” Hari writes.) For Super Bowl parties, Jackson’s avocado oil potato chips rather than Lay’s. Looking for a less processed alternative to Chick-fil-A’s frosted lemonade? Why not make your own with lemon-flavored protein powder from Hari’s own brand, Truvani. At least one attempt at a healthy food swap struck out with Hari: PepsiCo’s recently debuted dye-free line of Cheetos and Doritos. “This is dumb,” she wrote on Instagram. “Creating a whole NEW product, instead of FIXING their old product.”
Though the vast majority — 84% — of Americans said eating healthfully was at least moderately important to them in a recent Deloitte survey, most admit their own habits fall short of their aspirations. The $156 billion packaged snack industry has spotted a business opportunity in catering to people seeking a more enlightened way of noshing.
Opinion: NASA’s plans for living in space should prioritize immunology and infectious disease
In the aftermath of the successful Artemis II mission, NASA is moving forward with the next steps of its plans to establish a base on the moon. According to NASA Administrator Jared Isaacman, crews will be operating at the lunar base within the next decade with an even more ambitious long-term goal: Mars.
Human health in the space environment will be an important factor in these efforts. Among the concerns NASA should consider is the potential impacts of immunology and infectious disease.
Opinion: The CDC’s ‘disease detective’ program turns 75
The phone rings on the evening of Feb. 28, 2020.
“We need you to deploy to Seattle. Meet your team at Roybal tomorrow and additional details will be provided.”
STAT+: At AACR, more strong results for Revolution Medicine’s KRAS drug, plus assurance from NCI’s director
You’re reading the web version of STAT’s popup newsletter, AACR in 30 seconds, your guide to what’s happening at the American Association of Cancer Researchers’ annual meeting. Sign up here.
We’re nearing the end of a big AACR. We hope to see everyone at our live event on Tuesday night. Clearly, Revolution Medicines and KRAS have been the big topic of the meeting. Last year, AACR was dominated by big concerns over what cancer research funding would look like in the Trump administration. This year, the new head of the NCI tried to allay researchers’ fears. Read on!
Strong results for Revolution Medicines’ KRAS drug
Last week, researchers working with the biotechnology firm Revolution Medicines presented stunning news: the experimental drug daraxonrasib more than doubled survival in second-line pancreatic cancer compared to chemo — although that only meant increasing median survival in this terrible disease by six months.
STAT+: Kyverna Therapeutics plans to submit cell therapy for stiff person syndrome for FDA approval
A one-time, personalized cell therapy from Kyverna Therapeutics improved mobility and reduced disabilities in patients with stiff person syndrome, a rare, neurological autoimmune disorder, according to study results presented Tuesday.
Kyverna intends to submit the treatment to the Food and Drug Administration by the middle of the year. If approved, it would become the first treatment for stiff person syndrome and the first personalized CAR-T therapy for an autoimmune disease of any kind to reach the market.
Currently, CAR-T treatments are approved only for blood cancers, but using engineered T cells to deplete B cells — essentially performing an immune system reset inside a patient — has pushed a growing number of biotech companies to shift their CAR-T focus to autoimmune diseases.
STAT+: From Revolution Medicines, more strong data on KRAS drug and a glimpse of a ‘novel class’ beyond it
SAN DIEGO — Revolution Medicines is already cooking up the next iteration of RAS inhibiting drugs.
At the American Association of Cancer Research annual meeting here, the company is the talk of the town for the clinical trial success of daraxonrasib, its next generation targeted therapy, in advanced pancreatic cancer. And while the company presented more data on that drug Tuesday, showing promising first line and combination data on daraxonrasib, scientists also showed in another session intriguing preclinical data on a completely new compound that may represent what comes after the current lineup.
That drug, currently called RM-055, is what RevMed CEO Mark Goldsmith is calling an entirely “novel class of catalytic inhibitors.” These are targeted therapies that not only block the RAS signaling that drives cancer, but molecularly turn the cancer protein off.
STAT+: With successful trials, Roche takes its MS drug to regulators, but safety questions loom
The Swiss drugmaker Roche on Tuesday presented the latest data for its experimental multiple sclerosis drug, setting the stage for the company to seek approval for a medicine that it believes can cut relapse rates and slow the progressive disability the disease causes.
Now the test is whether the drug, called fenebrutinib, can win the regulatory green light.
While three late-stage trials of the drug have shown it to be effective, analysts have homed in on some potentially worrying liver safety signals, an issue that previously prompted the Food and Drug Administration to reject an MS therapy developed by Sanofi. In data released Tuesday, researchers also disclosed that there were two drug-related deaths among patients who took fenebrutinib.
Judge postpones OxyContin-maker Purdue Pharma’s sentencing to let opioid victims attend in person
A judge on Tuesday delayed the criminal sentencing of OxyContin maker Purdue Pharma in order to allow victims to attend the court proceeding in person.
U.S. District Judge Madeline Cox Arleo originally planned to hand down the sentence Tuesday during a court hearing conducted only by videoconferencing. But she said she changed her mind after seeing some victims of the opioid crisis protesting outside her courthouse in Newark, New Jersey. She said they should be allowed to attend in person, too, and moved the hearing to next Tuesday.
STAT+: Insurers refuse to join Medicare pilot offering weight loss drugs to seniors at steep discount
WASHINGTON — The Trump administration had planned for insurers to provide cheap weight loss drugs to seniors through a Medicare pilot program. Insurers said no, so the government will instead cover the drugs outside of the Medicare Part D drug benefit.
Last year, the Trump administration struck a deal with Eli Lilly and Novo Nordisk to lower the prices of their popular obesity drugs in exchange for expanding access to those drugs in Medicare and Medicaid. Medicare is prohibited from covering weight loss drugs, but the administration proposed waiving that restriction to test whether covering the medications would save tmoney or improve health outcomes at no additional cost.
The hitch was that insurers had to agree to participate in the pilot, called BALANCE, even though they would likely have faced a financial strain from doing so. The drugmakers agreed to sell the treatments for $245 a month in Medicare and Medicaid, but Medicare promised that beneficiaries would only pay $50 each month.
STAT+: Key GOP senators push back on Trump’s plan to cut NIH, reorganize HHS
WASHINGTON — Both Democratic and Republican senators who oversee federal spending seemed skeptical of proposed cuts to health research and public health in the White House’s budget, potentially teeing up a congressional package that ignores many of the administration’s most dramatic proposals for a second year.
During a Senate appropriations health subcommittee hearing Tuesday, lawmakers questioned health secretary Robert F. Kennedy Jr. on how his department could tackle chronic disease, smoking cessation, and cancer research with a proposed 2027 fiscal budget that would cut the department by 12%.
The request, which is broadly similar to what was proposed last year, includes deep cuts to the National Institutes of Health, the elimination of a health research agency, and the creation of a new agency devoted to chronic diseases called the Administration for a Healthy America.
STAT+: Key GOP senators push back on Trump’s plan to cut NIH, reorganize HHS
WASHINGTON — Both Democratic and Republican senators who oversee federal spending seemed skeptical of proposed cuts to health research and public health in the White House’s budget, potentially teeing up a congressional package that ignores many of the administration’s most dramatic proposals for a second year.
During a Senate appropriations health subcommittee hearing Tuesday, lawmakers questioned health secretary Robert F. Kennedy Jr. on how his department could tackle chronic disease, smoking cessation, and cancer research with a proposed 2027 fiscal budget that would cut the department by 12%.
The request, which is broadly similar to what was proposed last year, includes deep cuts to the National Institutes of Health, the elimination of a health research agency, and the creation of a new agency devoted to chronic diseases called the Administration for a Healthy America.
STAT+: CMS to ask every state for new audits of health care providers
WASHINGTON — The Trump administration will ask states to create new plans to verify medical providers paid by federally funded health care programs as part of a broad effort to combat fraud in government programs, Mehmet Oz, the administrator of the Centers for Medicare and Medicaid Services, said Tuesday.
The administration will ask states to develop plans in the next month to revalidate providers in “high-risk” areas to determine whether they exist and have the right to provide services, Oz said at Politico’s health care summit.
The announcement is another step from the administration in its aggressive — and highly publicized — attacks on alleged waste, fraud, and abuse in federal health care programs. The administration has targeted certain programs and providers primarily in Democrat-led states such as Minnesota and California, though the administration’s claims have been disputed at times by state leaders.
Opinion: STAT+: I’m an expert on presidential health. The 25th Amendment is not an option for removing Trump
Noting President Trump’s recent erratic public behavior, scores of legislators and political commentators have urged invoking the 25th Amendment to replace him with Vice President JD Vance. These individuals, largely Democrats, have questioned the president’s mental health, calling him “insane,” “deranged,” “unhinged,” and “batshit crazy,” among other terms. In response, the White House continues to defend Trump’s behavior as keeping his perceived opponents on edge.
Those calling for Trump’s removal under the 25th Amendment are misguided. Here’s why.
The 25th Amendment, which became law in 1967, is designed to ensure a prompt, orderly transfer of power if the president dies or becomes ill. Under the amendment’s terms, the vice president is indispensable in activating it, and involuntarily removing the president would need the support of a majority of the Cabinet and a two-thirds vote of both houses of Congress.
STAT+: RFK Jr. to face four GOP doctors at Senate hearings
You’re reading the web edition of D.C. Diagnosis, STAT’s twice-weekly newsletter about the politics and policy of health and medicine. Sign up here to receive it in your inbox on Tuesdays and Thursdays.
For National Library Week, STAT is offering 60% off subscriptions. Send news tips and fiction suggestions to John.Wilkerson@statnews.com or John_Wilkerson.07 on Signal.
The Senate GOP doctors who will question RFK Jr.
There are four Republican doctors on two of the Senate committees at which health secretary Robert F. Kennedy Jr. will testify this week. Two have been critical of Kennedy’s anti-vaccine policies, and we’ll be watching how they question the secretary, especially because the Republican party is trying its best to avoid talking about vaccines in the lead-up to the midterm elections.
STAT+: UnitedHealth earnings beat expectations in the first quarter, but hurdles persist
UnitedHealth Group, a company defined by growth, is systematically paring back parts of its business it spent years building up: Medicare Advantage and its sprawling provider network. That retreat is now paying off.
UnitedHealth’s financial performance in the first quarter, released Tuesday, far exceeded analysts’ expectations in both its Optum and UnitedHealthcare divisions. As a result, the company raised its full-year adjusted earnings outlook by 2.8% to $18.25 per share. Analysts praised the company’s performance in their notes, and UnitedHealth’s stock climbed as much as 10% mid-morning, even giving its competitors a lift.
Leaders who spoke on the company’s earnings call Tuesday morning sounded celebratory yet cautious, noting that both its insurance division, UnitedHealthcare, and its provider division, Optum Health, accrue most of their earnings in the first half of the year.
Flu vaccine no longer mandated for U.S. troops, defense secretary says
WASHINGTON — Defense Secretary Pete Hegseth announced Tuesday that the U.S. military will no longer require all American troops to get the flu vaccine, citing “medical autonomy” and religious freedom.
“The notion that a flu vaccine must be mandatory for every service member, everywhere, in every circumstance at all times is just overly broad and not rational,” Hegseth said in a video posted on social media.
Providers push back on 340B rebate model
UnitedHealth Reports Strong Quarterly Turnaround, Raises Full-Year Outlook
UnitedHealth Group delivered stronger-than-expected first-quarter results and lifted its full-year guidance, signaling momentum in its turnaround strategy and boosting investor confidence following last year’s earnings disappointment.
The healthcare giant reported adjusted earnings of $7.23 per share, surpassing analyst expectations of $6.58, according to FactSet data. Net income came in at $6.48 billion, or $6.90 per share, compared with $6.47 billion, or $6.85 per share, in the same period a year earlier. Revenue rose to $111.7 billion, up from $109.6 billion, reflecting steady growth across its insurance and healthcare services businesses.
“The results suggest meaningful progress in execution,” said Kevin Fischbeck, Managing Director and Senior Healthcare Analyst at Bank of America, noting that margin discipline and operational improvements are beginning to translate into earnings upside.
The company also raised its full-year adjusted earnings guidance by 50 cents to more than $18.25 per share, exceeding the FactSet consensus estimate of $17.86. The upgraded outlook points to improving visibility into cost management and underlying demand trends.
“Guidance increases of this magnitude typically indicate confidence in both cost control and revenue stability,” said Lisa Gill, Healthcare Services Analyst at JPMorgan, adding that the company appears to be regaining footing after a volatile prior year.
The performance marks a notable shift from last year, when an unexpected earnings shortfall triggered a sharp selloff in UnitedHealth shares. The company has since undergone significant leadership and operational changes under Chief Executive Stephen Hemsley, who returned to the role in May with a mandate to reset the business.
Hemsley has focused on restructuring operations, tightening cost controls, and improving margins. “Turnarounds of this scale require both leadership change and execution discipline,” said Ricky Goldwasser, Healthcare Analyst at Morgan Stanley, highlighting that management actions are now beginning to show measurable impact.
As part of its strategic overhaul, UnitedHealth said it has replaced nearly half of its top 100 executives and accelerated investments in artificial intelligence to improve efficiency and healthcare cost management. The company also announced an agreement to acquire Alegeus Technologies, a benefits administration firm, though financial terms were not disclosed.
“Technology investment, particularly in AI, is becoming central to managing medical costs,” said David Windley, Healthcare Analyst at Jefferies, noting that digital tools are increasingly driving competitive advantage in managed care.
A key metric for investors—the medical-loss ratio (MLR)—came in at 83.9%, significantly below analyst expectations of 85.5%. The ratio, which measures the percentage of premium revenue spent on healthcare costs, is closely watched as an indicator of profitability.
“The lower MLR is a strong positive signal,” said Sarah James, Healthcare Analyst at Cantor Fitzgerald, adding that improved cost management and reserve development played a critical role in the outperformance.
UnitedHealth said the lower ratio was driven by “strong medical cost management and favorable reserve development,” even as overall healthcare costs remain elevated across the industry.
The results come amid a more supportive backdrop for insurers, following a recent announcement that Medicare reimbursement rates for 2027 would come in higher than initially proposed. The development has lifted sentiment across the managed-care sector.
“Rate visibility is critical for the group,” said Josh Raskin, Senior Healthcare Analyst at Nephron Research, noting that improved reimbursement outlooks provide a clearer earnings runway.
UnitedHealth, the parent of the largest U.S. health insurer, continues to benefit from its diversified model, which includes insurance, healthcare services, and physician networks. That structure allows it to manage costs more effectively than peers during periods of volatility.
Looking ahead, investors will be watching whether the company can sustain its margin improvements and continue executing on its restructuring plan. With stronger guidance, improved cost metrics, and leadership stability, UnitedHealth appears to be regaining momentum—but the durability of the turnaround will remain the key question for markets.
JBizNews Desk
STAT+: Pharmalittle: We’re reading about a Purdue Pharma settlement, a setback for Merck and Eisai, and more
Top of the morning to you, and a fine one it is. Sunny skies and mild breezes are enveloping the Pharmalot campus once again. And to celebrate, we are brewing another cuppa stimulation and inviting you to join us. Our choice today is blackberry hibiscus. Remember, a prescription is not required. So no need to mess with rebates or even TrumpRx. Meanwhile, here are a few items of interest. Hope you have a meaningful and productive day and, of course, do stay in touch. …
Pfizer executive Andrew Baum, a former analyst at Citibank who joined the drugmaker in June 2024 to redirect its strategic approach, has left his role as an executive vice president and chief strategy and innovation officer, STAT writes. He will continue as an adviser to Pfizer chief executive officer Albert Bourla until the end of the year. Baum was part of a mini-trend of large pharmaceutical companies hiring former analysts at investment banks, who closely track multiple companies in order to help investors understand their businesses, as strategic leaders. Ronny Gal, a longtime analyst at Bernstein, became chief strategy and growth officer at Novartis in 2022. Christopher Shibutani joined Bristol Myers Squibb as chief strategy officer last year.
A U.S. judge is expected to sentence OxyContin maker Purdue Pharma to forfeit $225 million to the U.S. Department of Justice on Tuesday, clearing the way for the company to finalize a settlement of thousands of lawsuits it faces over its role in the opioid crisis, the Associated Press tells us. The penalty was agreed to in a 2020 pact to resolve federal civil and criminal probes it was facing. If the judge signs off, other penalties will not be collected in return for Purdue settling the other lawsuits. The settlement was approved by another judge last year and could take effect on May 1. It requires members of the Sackler family who own the company to pay up to $7 billion to state, local, and Native American tribal governments, some individual victims, and others.
STAT+: Kelonia Therapeutics’ road from floundering to $3 billion
Want to stay on top of the science and politics driving biotech today? Sign up to get our biotech newsletter in your inbox.
Good morning. Starting and sustaining a biotech company is never easy. If you need a reminder on that, read to the end of today’s newsletter.
BioAge’s bet on inflammation shows early promise
BioAge is making its way into the race among drug companies to target inflammation as a way to treat chronic conditions.
This CDC conference was always open to the public. Until now
Get your daily dose of health and medicine every weekday with STAT’s free newsletter Morning Rounds. Sign up here.
Good morning. I have a confession: Yesterday was the first time I ever logged into my patient portal to check my medical records. Seeing this in a clinical note from a few years ago made me chuckle: “Lives alone with cat.”
STAT+: Kennedy hints at coming breakthrough device announcement
You’re reading the web edition of STAT’s Health Tech newsletter, our guide to how technology is transforming the life sciences. Sign up to get it delivered in your inbox every Tuesday and Thursday.
Good morning health tech readers!
Maybe the Trump administration has grand plans to cover breakthrough devices after all?
STAT+: BioAge says experimental pill aimed at reducing heart risks significantly reduced inflammation
BioAge Labs said Tuesday that its investigational pill for cardiovascular risk prevention significantly reduced inflammation in an early study, as more drug companies target inflammation as a way to treat a range of chronic conditions.
In a Phase 1 study of people with obesity and elevated inflammation levels, patients taking a 60-milligram dose of the drug, called BGE-102, experienced an 86% reduction in a measure of inflammation called high-sensitivity C-reactive protein (hs-CRP) after three weeks. That’s a similar level of reduction seen in patients who took a higher 120-mg dose in the study, which the company previously reported.
Additionally, 87% of patients taking the 60-mg dose achieved hs-CRP levels of less than 2 mg/liter, the threshold thought to be associated with a lower risk of cardiovascular complications.
STAT+: Tortugas Neuroscience launches with hopes to develop drugs for brain disorders, other conditions
A new startup has found that the Chinese biotech industry is good for more than obesity drugs and cancer therapies.
The company, Tortugas Neuroscience, launched Tuesday with plans to develop two schizophrenia and tinnitus drugs licensed from Chinese drugmaker Jiangsu Hansoh Pharmaceutical Group. The startup will also test two other medicines for focal epilepsy and encephalopathies that were originally created by Japanese pharmaceutical company Eisai Co. Ltd.
Tortugas has $106 million from Cure Ventures, The Column Group, and AN Ventures to begin testing the drugs in mid-stage trials in the U.S.
Opinion: What happens when a chief executive loses executive functions?
Circa 1970, the renowned Russian neuropsychologist Alexander Luria together with Karl Pribram from Stanford University and other neuroscientists of that era introduced the term “executive functions” into the scientific lexicon to denote complex behaviors such as attention and awareness. They identified the frontal lobe — the front of the brain — as the “executive of the brain” responsible for these behaviors based on their experiments with primates and patients with specific brain injuries.
Over time, the concept evolved to include mental processes needed to focus, concentrate, and pay attention when challenged by multiple simultaneous sources of information to weigh options and make informed decisions as opposed to impulsive ones.
Opinion: The U.S. health care system is built around looking backward
In outpatient medicine, decisions move quickly.
A prescription is sent to a pharmacy. A referral goes out to a specialist. A test is ordered. From the patient’s perspective, the process feels immediate. Care begins moving forward as soon as the visit ends.
Trump Orders New Tariffs on Imported Drugs, Escalating Push to Bring Pharmaceutical Manufacturing Back to the U.S.
The Trump administration on Monday moved to impose a new round of tariffs on imported pharmaceutical products, marking one of the most aggressive steps yet to pressure drugmakers to relocate manufacturing operations to the United States, according to senior administration officials familiar with the policy. President Donald Trump said at a White House briefing the move is designed to end what he called “dangerous dependence” on foreign drug supply chains, particularly from China and India, which together account for a significant share of active pharmaceutical ingredient (API) production used by U.S. manufacturers.
Commerce Secretary Howard Lutnick said the tariffs will apply to a range of finished drugs and key ingredients, with rollout expected in phases over the coming months. “The United States cannot remain reliant on overseas production for critical medicines,” Lutnick said in a statement issued by the Department of Commerce. “This is about national security, supply chain resilience, and restoring high-value manufacturing jobs.” The department pointed to lessons from COVID-era shortages, when overseas disruptions exposed vulnerabilities in essential drug supplies.
The action is being structured under authorities similar to Section 232 of the Trade Expansion Act, which allows tariffs on imports deemed a national security risk. U.S. Trade Representative Jamieson Greer said the administration is aligning trade policy with industrial strategy. “We are sending a clear message to global manufacturers: access to the U.S. market comes with expectations around supply chain security,” Greer said during a policy briefing, according to an official transcript released by the Office of the USTR.
Industry groups pushed back, warning the policy could raise costs and strain already fragile supply chains. Stephen J. Ubl, President and CEO of the Pharmaceutical Research and Manufacturers of America (PhRMA), said “while strengthening domestic manufacturing is a shared goal, tariffs risk increasing costs for patients and could lead to unintended supply disruptions.” PhRMA has consistently advocated for tax incentives and regulatory streamlining as more effective tools to drive U.S.-based production.
Health policy experts say the impact may be most acute in the generic drug market. Dr. Marta Wosińska, Senior Fellow at the Brookings Institution and former FDA chief economist, noted that many generic manufacturers operate on razor-thin margins. “Even modest tariffs could push some suppliers out of the market, increasing the risk of shortages,” she said in a recent Brookings analysis, emphasizing that domestic capacity cannot be scaled overnight.
The administration argues the long-term strategic benefits outweigh near-term disruption. National Security Advisor Robert O’Brien said the policy is part of a broader effort to secure critical industries. “We cannot afford to rely on geopolitical competitors for lifesaving medications,” O’Brien said in a televised interview. “This is about ensuring supply continuity in times of crisis.”
Data from the U.S. Food and Drug Administration (FDA) shows a large share of APIs used in U.S. drugs are manufactured abroad, with China and India dominating production. The agency has repeatedly highlighted challenges in monitoring overseas facilities, reinforcing concerns among lawmakers about quality control and supply chain concentration risks.
On Capitol Hill, reaction has split along strategic and economic lines. Sen. Marco Rubio (R-Fla.) said in a statement that reshoring pharmaceutical manufacturing is “essential to America’s national and economic security.” Meanwhile, Sen. Ron Wyden (D-Ore.), Chairman of the Senate Finance Committee, warned that “any tariff policy must include safeguards to prevent higher prescription drug costs for American families,” according to remarks released by his office.
Major pharmaceutical companies including Pfizer, Merck, and Johnson & Johnson have already begun expanding U.S.-based manufacturing in recent years, though industry analysts note global supply chains remain deeply embedded overseas due to cost advantages and existing infrastructure.
What comes next will depend on execution. Analysts say the success of the policy will hinge on whether tariffs are paired with incentives, workforce investment, and regulatory reforms to make domestic production economically viable at scale. Without that, higher costs could ripple through insurers, hospitals, and ultimately consumers, even as supply chains adjust.
As the policy moves toward implementation, the global pharmaceutical industry is bracing for a significant realignment—one that could reshape where and how critical medicines are produced, with lasting implications for trade, healthcare costs, and national security.
— JBizNews Desk
STAT+: At AACR, a provocative use of CAR-T, Merck’s new thing and cancer’s geography problem
You’re reading the web version of STAT’s popup newsletter, AACR in 30 seconds, your guide to what’s happening at the American Association of Cancer Researchers’ annual meeting. Sign up here.
We’re back with more data from the AACR meeting! Among the highlights today: a first look at a drug Merck acquired from China, a fascinating but potentially controversial use for CAR-T, and American oncology’s geography problem. Don’t forget: Tuesday night we will host a live event in San Diego, and we also have a virtual recap of the AACR conference on Thursday.
CAR-T shows deep response in smoldering myeloma
In an early phase trial, investigators at Dana-Farber Cancer Institute treated 20 high risk smoldering multiple myeloma patients with Carvykti, a BCMA directed CAR-T therapy. The idea was to use the immunotherapy on patients with the multiple myeloma precursor condition, hoping to prevent the active cancer in patients at high risk of progression.
STAT+: Former banker brought on to revitalize Pfizer’s strategy departs his post
Pfizer executive Andrew Baum, a former analyst at the investment bank Citi who joined the company in June 2024 to redirect its strategic approach, has left his role as an executive vice president and as chief strategy and innovation officer, the company confirmed Monday. He will continue as an adviser to Pfizer CEO Albert Bourla until the end of the year.
Pfizer and sources familiar with the matter described the move as stemming from both a sense that Baum had accomplished what he set out to do and a streamlining of Pfizer’s operations.
“Pfizer regularly evaluates its operations to ensure it is best positioned to deliver on the company’s business in the near-term and beyond,” the company said in a statement. It said that organizational and leadership changes would “position Pfizer to move faster, make clearer decisions, and advance innovation across the enterprise.”
STAT+: In early trial, CAR-T results raise hope of preventing multiple myeloma in high-risk patients
SAN DIEGO — Alison Cameron spent close to a decade fighting to keep myeloma under control. She’d been diagnosed with smoldering multiple myeloma, a precursor to cancer, and spent years receiving infusions to keep it from progressing to active multiple myeloma. Now, after receiving CAR-T therapy, an aggressive immunotherapy, while on a trial, the 54-year old anesthesiologist is hoping the risk of cancer is gone for good.
It’s a reasonable hope, given the results of that trial, which researchers presented at the American Association for Cancer Research meeting here on Monday. All 20 patients who received the trial treatment no longer had any detectable myeloma cells in their body. That’s a far deeper and more complete response than scientists typically expect when it comes to multiple myeloma, and it’s prompting some experts to consider the possibility these patients have truly had active cancer permanently averted.
Currently, there is only one approved therapy for high risk smoldering myeloma, an antibody therapy called Darzalex. Patients can remain on treatment for years, but without achieving these kinds of deep molecular responses, and many still progress within 5 years, said Ecaterina Dumbrava, a cancer researcher at MD Anderson Cancer Center who did not work on the study. “The results raise a very important question whether early immune interception can not only delay progression but redefine treatment goals. Can we talk about the word we always avoid, which is cure,” she said.
Opinion: The contradiction at the heart of Republicans’ embrace of psychedelics
On Saturday, with podcaster and psychedelics fan Joe Rogan standing over his shoulder, President Trump signed an executive order intended to expand access to psychedelics for mental health treatment.
For some, it may come as a surprise to see a Republican president embrace psychedelics. But he’s only the most recent member of the GOP to hop on the psychedelics bandwagon.
STAT+: Trump order to advance psychedelic treatments generates excitement — and worries
President Trump’s executive order aimed at loosening restrictions on psychedelics as mental health treatments was largely applauded by advocates. But some also quietly worry the White House’s role in trying to bolster the field risks politicizing it and undermining the credibility of research.
The order, which Trump said originated with a text from podcaster Joe Rogan about psychedelics research, directs the Food and Drug Administration to expedite the review of some compounds and calls for the establishment of a new regulatory pathway for experimental psychedelics to be tried by terminally ill patients. It also allocates funding to states developing research programs.
While the order does not actually reschedule any drugs or change legislation, many advocates and researchers welcomed the move, saying it signals the administration’s interest in advancing psychedelics as treatments and could help ease bottlenecks in expanding access.
STAT+: Eli Lilly’s $3.25 billion acquisition of Kelonia Therapeutics caps startup’s tortuous ride
Eli Lilly is spending $3.25 billion to acquire Kelonia Therapeutics, a small biotech company developing cell therapies for cancer and autoimmune diseases, the companies announced Monday.
Lilly could pay more if Kelonia achieves specified clinical, regulatory, and commercial milestones.
The acquisition is a boon for the small startup, which has subsisted on just $60 million over the last five years and previously struggled to stay afloat. Three times, the company came within a week of running out of cash, according to Bryan Roberts, a partner at the venture capital firm Venrock, which incubated the biotech.
STAT+: The PBMs fight back, and arbitration doesn’t lose
This is the online version of STAT’s weekly email newsletter Health Care Inc. Sign up here.
Did you know it’s National Library Week? Of course you did, you look so studious. To honor the thrill of reading and learning, STAT is offering 60% off our subscription for your first year. Check it out, visit a library, and as always, say hello: bob.herman@statnews.com.
A contentious fight over drug price transparency
Anna Yeo/STAT
The phalanx of lobbyists representing pharmacy benefit managers and health insurers has mounted stiff opposition to a federal proposal that attempts to pry open the black box of prescription drug prices.
STAT+: Finally cracking KRAS as a druggable target
Want to stay on top of the science and politics driving biotech today? Sign up to get our biotech newsletter in your inbox.
Hiya. Today, we see a proliferation of KRAS, as new data validate a once-undruggable space. Also, a double-edged push for PBM transparency, and the Breakthrough Prize was awarded to researchers for advances in gene therapy and ALS genetics.
The need-to-know this morning
- Novo Nordisk said its experimental oral drug, called etavopivat, reduced the risk of pain crises and improved hemoglobin response in patients with sickle cell disease, achieving the goals of a Phase 3 clinical trial.
- Eli Lilly is reportedly nearing a deal to acquire Kelonia Therapeutics for more than $2 billion, according to the Wall Street Journal. Kelonia is a privately held developer of so-called in-vivo CAR-T therapies that make engineered cancer-killing cells inside the body. Its lead therapy is being investigated in an early stage study in multiple myeloma.
- Helus Pharma, developer of psychedelics-based therapies for depression, said CEO Michael Cola has resigned “at the request of the board of directors.” Chairman Eric So was appointed interim CEO.
Finally cracking KRAS as a druggable target
When Revolution Medicines released results for its drug daraxonrasib in pancreatic cancer patients last week, it finally seemed like there was hope in a condition that has long proven intractable for researchers. But as STAT’s Angus Chen reports, it was the product of years of effort — and could be just the start of a wave of new RAS inhibitors.
STAT+: Pharmalittle: We’re reading about Trump boosting psychedelic treatment, the future for weight-loss drugs, and more
Good morning, everyone, and welcome to another working week. We hope the weekend respite was relaxing and rewarding, because that oh-so-familiar routine of meetings, online calls, and deadlines has predictably returned. But what can you do? The world keeps spinning no matter how much we push back. So time to get on with it. Please join us for a cup of stimulation — hot buttered rum is our choice today — as we cast about for items of interest. Speaking of which, here are some tidbits. Hope your day goes well…
Weight-loss drugs may eventually reach only about 50% of the overweight and obese people who could benefit from them due to complexities of health care systems and financial constraints, Reuters writes, citing comments made by Eli Lilly chief executive officer David Ricks said at a conference. Lilly and Novo Nordisk are competing for share in the global market for GLP-1 drugs, which analysts see topping $100 billion per year in the next decade. But just one in 10 people who are overweight or obese are using GLP-1s. “It’s never going to be a hundred,” Ricks said. “For institutional reasons in health care and some other complexities in managing health, it’s never going to be that high.” He pointed to low-cost statins, the most commonly prescribed cholesterol drugs, as a comparable example. “Between 40 and 50% of people who should be on them, are on them. I think of that as maybe a ceiling,” he said.
The Trump administration’s desire to pry open the black box of prescription drug prices is facing stiff opposition from the phalanx of lobbyists representing pharmacy benefit managers and health insurers, STAT notes. In January, the U.S. Department of Labor proposed a rule that would mandate PBMs disclose a wide range of drug pricing information to employers and make it easier to be audited. The public had until last week to submit comments. A review of the more than 500 letters reflects varying interests: predictable resistance from PBMs and health insurers; enthusiasm from Mark Cuban’s pharmacy and others in the business community who want middlemen to face more accountability; and drug companies cheered PBMs being in the regulatory crosshairs but wanted pullback on disclosure of drug pricing data.
STAT+: Extended use of Nektar Therapeutics drug shows promise in alopecia
Nektar Therapeutics said Monday that extended treatment with its experimental drug, called rezpeg, promoted greater hair growth in people with severe alopecia areata, an autoimmune condition that attacks hair follicles.
After one year, 27% of participants treated with either a low or high dose of rezpeg achieved a clinically meaningful hair-growth outcome, Nektar said. The response, formally known as SALT Score 20, is defined as 80% or more of the scalp covered by hair.
The rezpeg results are equal or better to those from a low dose of Olumiant, a daily pill made by Eli Lilly that is currently approved for severe alopecia areata, albeit with physician use constrained by safety concerns. The two drugs have not been tested head to head.
How weed affects adolescent brains
Get your daily dose of health and medicine every weekday with STAT’s free newsletter Morning Rounds. Sign up here.
Good morning and happy Marathon Monday from Boston! If you’ve been waiting for the right deal to finally sign up for STAT+, then you’re in luck. This week, we’re running what STAT newsletter strategist Alexa Lee calls “one of the fattest discounts we’ve done in a while!” Get it while the getting is good.
3 ways AI is humanizing patient care
STAT+: A controversy over research monkeys highlights ambiguity over health standards
A leading animal rights group is accusing Pfizer of running afoul of its own standards in the handling of research monkeys, jeopardizing both the welfare of the animals and the integrity of scientific research.
But the case also appears to highlight a lack of guidelines for assessing the health of monkeys before they are shipped for medical research.
At issue are nearly three dozen monkeys that Pfizer procured in late 2024 from an academic research center before making plans to have them shipped to a contract research organization for clinical work.
Opinion: Politicians are using low teen birth rates to further restrict access to birth control, abortion
Recently, the Centers for Disease Control and Prevention published its most recent estimates of live births in the U.S. It showed that birth rates continue to decline nationwide, and teen birth rates specifically are at record lows. These findings have drawn widespread media attention and will certainly fuel the pronatalist movement’s concern that fewer people are getting married and having children. In fact, a senior medical analyst for Fox News explicitly described low teen birth rates as “the problem” driving overall fertility declines. He is not alone in this belief.
As a public health behavioral scientist who has spent my career researching adolescent access to sexual and reproductive health care, I felt a startling sense of whiplash upon hearing these panicked reactions. When I was at the CDC in the 2010s, during the “teen pregnancy prevention” era, these same data reports documenting declining teen birth rates were cause for celebration.
STAT+: PBMs warn Trump’s proposal to disclose drug prices is illegal
The Trump administration’s desire to pry open the black box of prescription drug prices is facing stiff opposition from the phalanx of lobbyists representing pharmacy benefit managers and health insurers.
In January, the Department of Labor proposed a rule that would mandate PBMs disclose a wide range of drug pricing information to employers and make it easier to be audited. The public had until last week to submit comments.
A review of the more than 500 letters that poured in reflects a melange of drug pricing interests: predictable resistance from PBMs and health insurers, which generate billions of dollars in profit every year from their role as gatekeepers of drug spending; enthusiasm from Mark Cuban’s pharmacy and others in the business community who want middlemen to face more accountability; and a semblance from the pharmaceutical industry of having one’s cake and eating it, too: Drug companies cheered PBMs being in the regulatory crosshairs but wanted pullback on disclosure of drug pricing data.
Opinion: Measles outbreak means my immunocompromised son can’t leave the house without extreme safety measures
My teenage son has Okur-Chung neurodevelopmental syndrome (OCNDS), an ultra-rare genetic disorder caused by a mutation on the CSNK2A1 gene, which creates the CK2 protein present in every cell in the body. Each patient is affected differently, but common symptoms include autism, intellectual disabilities, short stature, low muscle tone, and speech delay. Last year, he developed autoimmune encephalitis, which resulted in a two-month hospitalization. He is currently undergoing treatment that includes regular intravenous immunoglobulin infusions and a transplant rejection medication that severely suppresses the immune system.
Ever since the Covid pandemic began, my family has completely changed our lives to protect his health. I work remotely, foregoing work travel and in-person meetings to limit the chance of catching Covid or any other virus. My husband left the workforce to become a stay-at-home parent and homeschool our son to limit his exposure to illness. When we venture out to places like the local library, museum, or farm near our house, we stay safe by masking and social distancing.
Even for trans adults, care is hard to find: ‘I could not do it on my own’
SAVANNAH, Ga. — For Nikolas Indigo, the road to freedom was lined with warnings against a life of sin. Neither the road nor the message was a metaphor. Along nearly 250 miles of highway from Savannah to Atlanta, billboards preached: “turn from sin,” “Jesus is the way,” “repent.”
It’s a common pilgrimage for transgender people, who often need to travel for affirming procedures. Indigo, 25, made appointments with four different surgeons before he was finally able to get masculinizing chest surgery in Atlanta in September. Despite Savannah’s reputation for being young, hip, and at least a little queer, few local physicians perform the basic procedure.
STAT+: AACR 2026: More strong data for Revolution Medicines’ KRAS drug
You’re reading the web version of STAT’s popup newsletter, AACR in 30 seconds, your guide to what’s happening at the American Association of Cancer Researchers’ annual meeting. Sign up here.
We’re here in San Diego, on the ground at AACR — one of the best places to spot early cancer research that will turn out to be important later on. This year? Everything is coming up KRAS. And, STAT is here in force: Cancer reporter Angus Chen, reporter-at-large Damian Garde, and senior writer Matt Herper are all contributing, and we’ll be hosting an in-person event Tuesday and a virtual one Thursday. Sign up!
AACR leaders thank Congress for standing up for science
Last year, President Trump’s budgets included dramatic cuts to the National Institutes of Health, giving NIH funded scientists a scare and prompting outcry from scientific leaders including those at AACR. Congress firmly rejected those cuts last year, and increased the NIH budget for fiscal year 2026. In response, at this year’s AACR opening ceremony, AACR CEO Margaret Foti thanked Congress for standing up for science, with a callout for members of the Senate Appropriations Committee.
Earlier this month, Trump proposed a $5 billion cut to the NIH for 2027, which Foti called unacceptable during her opening remarks. “Our purpose is clear. Our mission is urgent. And our commitment is unwavering. We cannot allow our lifesaving mission to be adversely affected by the Administration’s plan to cut NIH funding by 20% for fiscal year 27,” she said.